---
Publish Now: null
author:
- Yujan Shrestha
cta_button_text: ''
cta_case_studies: []
cta_hero_details: ''
cta_hero_text: ''
date: '2026-05-06'
description: 27 AI/ML medical devices authorized in April 2026. Radiology\'s share
of clearances dropped from 79% to 56%, QIH remains the regulatory plumbing for image-processing
AI, and PCCP adoption is ticking up. A breakdown of what the April cohort reveals
about where AI/ML device strategy is heading.
related:
- 355bd5b7-a754-80fe-b92b-c8000a1b1a5c
- 2cebd5b7-a754-8038-9ed7-d6f99a7970e9
- 2e3bd5b7-a754-8062-bb08-f159e98f3580
- 2d5bd5b7-a754-80a4-ab9b-c8f2505ed08c
- 2cfbd5b7-a754-8059-b1cc-ffd20e4c8f47
- 18abd5b7-a754-8031-b9c5-ccafb5bb43e2
- db5cc020-6e32-4da6-9830-52a6094f26a6
- 7d4a0f52-d535-44cb-a51d-31414a8c270c
- 353bd5b7-a754-8055-8506-f19a9e8f91a1
title: 'April 2026: 26 AI FDA 510(k) and One De Novo'
topics:
- AI/ML
- News
- Regulatory
---
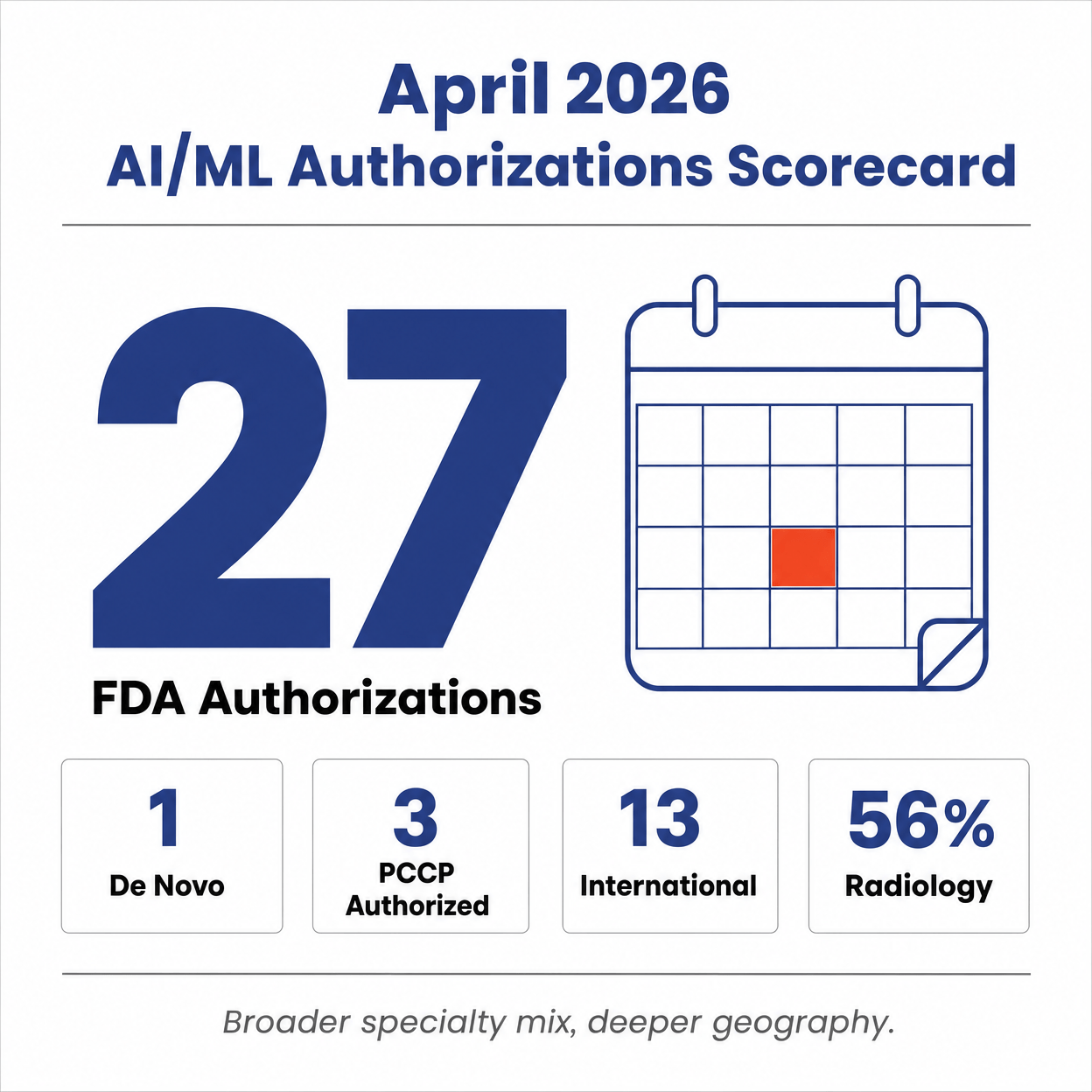
FDA authorized 27 unique AI/ML medical devices in April 2026. One
every 26.7 hours.
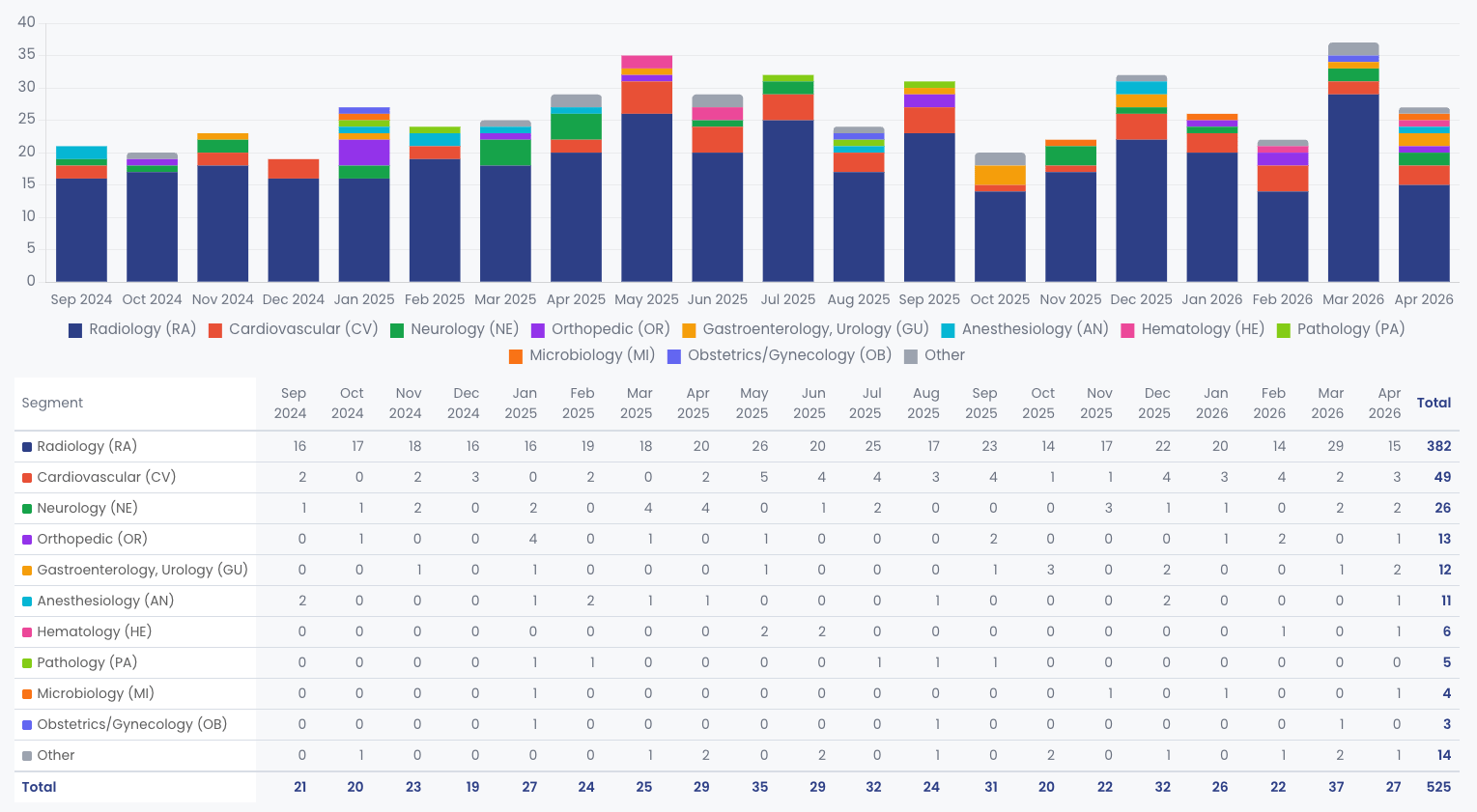
The FDA authorized 27 unique AI/ML-enabled medical devices in April 2026. That is one new AI device entering the US market every 26.7 hours.
While March was heavily dominated by standalone Software as a Medical Device (SaMD), April saw a broader mix. Of the 27 authorizations, 18 were SaMD, while the remaining 9 were AI-enabled hardware or in vitro diagnostic (IVD) systems. The cohort includes 26 510(k) clearances and one De Novo classification.
Here is a look at the trends driving the April 2026 cohort.
## Three new product codes established
April saw the creation of three new product codes for AI/ML devices:
SHO, SHP, and SHQ.
The FDA established three brand-new product codes for AI/ML devices in April 2026. While new codes are typically born out of De Novo classifications, this month proved that 510(k) clearances can also trigger new codes when they map to existing regulations.
The three new codes are:
1. SHO (Neurology): Established by Automated Imaging Diagnostics\' De Novo for \"neuropacs\" (DEN240071). This code covers diffusion MRI software intended to help differentiate Parkinsonian syndromes (like MSAp and PSP) from Parkinson\'s Disease. It was placed into Class II under 21 CFR 882.2000.
2. SHP (Cardiovascular): Established by Anumana\'s 510(k) for its ECG-AI Cardiac Amyloidosis algorithm (K253801). This code covers cardiovascular machine learning-based notification software under 21 CFR 870.2380 (a regulation originally established by the Viz HCM De Novo).
3. SHQ (Dental): Established by 3Shape\'s 510(k) for TRIOS Dx (K260082). This code covers AI-based dental diagnostic aids that analyze intraoral scans for caries, tooth wear, gingival recession, and plaque. It relies on the Dental Monitoring De Novo (DEN230035) as its predicate. Why a new product code? Because the existing dental ones were for xrays and CBCTs.
The creation of SHP and SHQ via the 510(k) pathway highlights an important regulatory nuance: once a pioneer clears the brush via a De Novo and establishes a regulation, fast-followers can secure 510(k) clearance while still prompting the FDA to carve out a specific new three-letter product code for their exact indication.
## Radiology still leads, but the specialty mix is broadening
Radiology accounted for 56% of April's AI authorizations, down from
79% in March.
Radiology remains the undisputed center of gravity for medical AI, but its share of the pie is shrinking as other specialties accelerate. In March, the Radiology panel reviewed a staggering 79% of all AI clearances. In April, that number dropped to 56% (15 of 27 devices).
The remaining 12 devices were spread across a diverse set of clinical areas. Cardiovascular led the non-radiology pack with three clearances, followed by Neurology, Gastroenterology/Urology, and Dental with two each. Microbiology, Orthopedics, and General Hospital each saw one clearance.
Notable non-radiology clearances included Bayesian Health\'s Sepsis Flagging Device (K250680), Wesper\'s CASSIE sleep apnea monitor (K252628), and bioMérieux\'s VITEK 2 AST-Streptococcus microbiology system (K260282). We also saw the first therapeutic AI device in recent months: Ottobock\'s myosmart prosthetic system (K253256), highlighting how machine learning is moving beyond diagnostics and into active patient intervention.
The De Novo standout: Neurological imaging
Just like in March, the FDA granted exactly one De Novo classification for an AI device in April.
Automated Imaging Diagnostics received De Novo authorization for \"neuropacs\" (DEN240071), an AI-powered neurological imaging tool. The FDA created a new product code (SHO) specifically for this device. This continues the trend of the agency using the De Novo pathway to establish regulatory guardrails for novel AI applications that lack a clear predicate.
## QIH remains the dominant regulatory plumbing
The QIH product code was used for 6 of the 27 clearances, spanning
diverse clinical applications.
If you want to understand how AI gets cleared, look at the product codes. In April, QIH (Automated Radiological Image Processing Software) was once again the most frequently used product code, attached to 6 of the 27 clearances.
What makes QIH fascinating is its anatomy-agnostic nature. In April alone, QIH was used to clear AI tools for dental X-rays (Overjet Iris), enteric tube placement (GE HealthCare), follicle scanning (MIM Fertility), dental CBCT (Dentsply Sirona), general radiology suites (Siemens Healthineers), and 3D imaging base tools (Fujifilm).
This confirms that QIH has become the default regulatory plumbing for any software that automates the processing of medical images, regardless of the specific body part being analyzed.
PCCP adoption ticks upward
Predetermined Change Control Plans (PCCPs) allow manufacturers to pre-clear specific future algorithm updates without needing a new 510(k). Adoption has been slow, but April showed a slight uptick.
Three of the 27 devices (11%) included an authorized PCCP, compared to just two in March. The April PCCP adopters were Anumana (ECG-AI Cardiac Amyloidosis), bioMérieux (VITEK 2 AST-Streptococcus), and Conavi Medical (Novasight Hybrid System). Notably, this is Anumana\'s second PCCP authorization in as many months, suggesting that some companies are building PCCPs into their standard regulatory playbook.
Geography and demographics
The globalization of medical AI continues. By our count, 13 of the 27 applicants (48%) are headquartered outside the United States. This includes companies from China (Shanghai United Imaging, Aitewan), Japan (Canon, Fujifilm), Germany (Siemens, Ottobock), Italy (Esaote), Denmark (3Shape), South Korea (Lunit), the Netherlands (Quantib), and Israel (Magentiq Eye).
On the demographic front, pediatric labeling remains relatively scarce. Only 4 of the 27 devices (15%) included pediatric labeling in their indications for use. These included United Imaging\'s uMI Panvivo, Esaote\'s MyLab E80 Ultrasound, GE HealthCare\'s Critical Care Suite, and Canon\'s Intelligent NR.
## Takeaway
April 2026 proves that the AI/ML device pipeline is not just growing in volume, but in breadth. While radiology and the QIH product code still provide the structural foundation for the industry, we are seeing meaningful expansion into cardiovascular, neurology, and even therapeutic applications like prosthetics.
As the specialty mix broadens, the regulatory strategies are maturing. The steady trickle of De Novos and the slight uptick in PCCP adoption indicate that both the FDA and manufacturers are getting more comfortable navigating the complexities of adaptive algorithms and novel clinical use cases.
*If you are building an AI/ML medical device and need help navigating the FDA\'s evolving expectations for PCCPs, clinical evaluation, or De Novo submissions, Innolitics can help. We specialize in software-as-a-medical-device engineering and regulatory affairs.*