About Innolitics 🔗
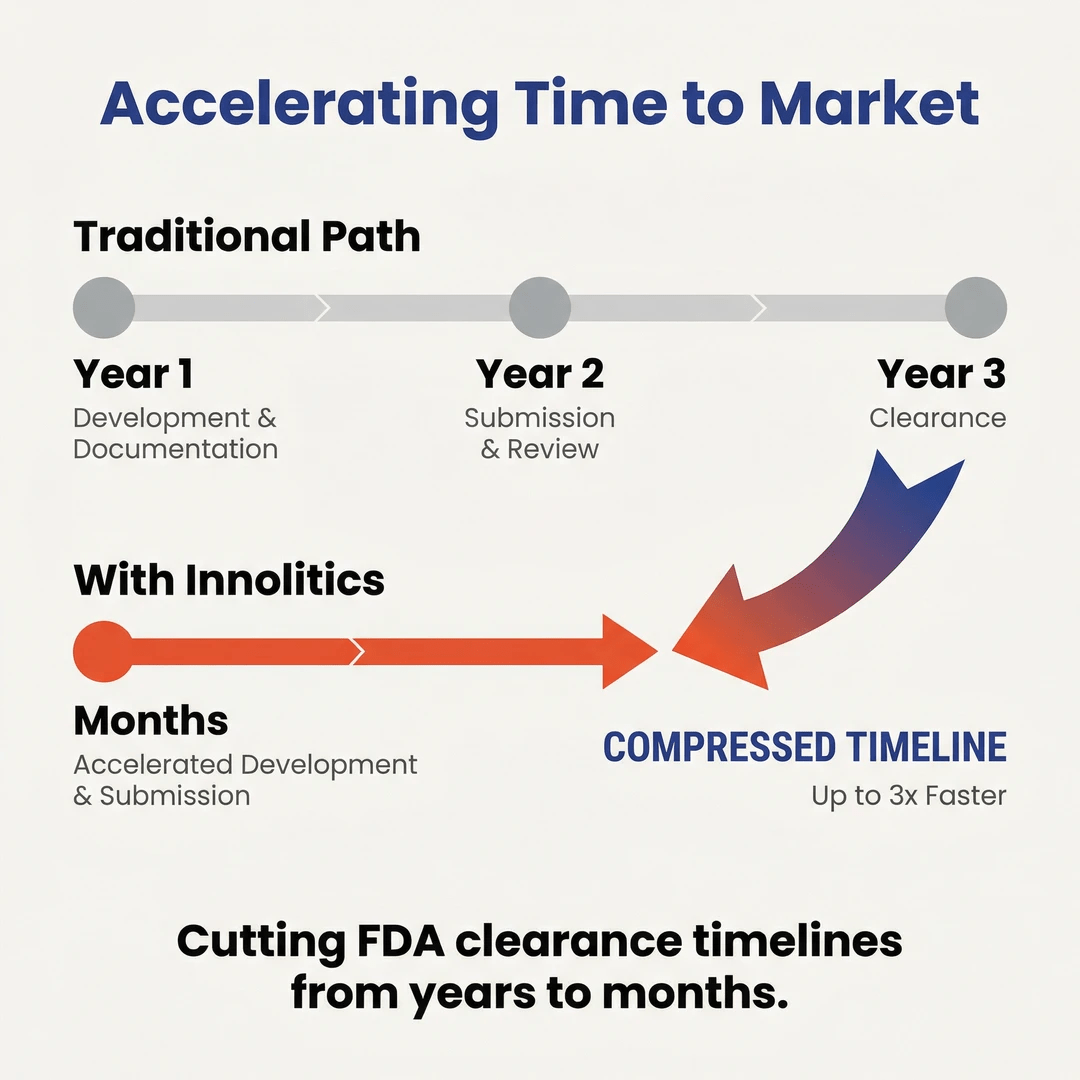
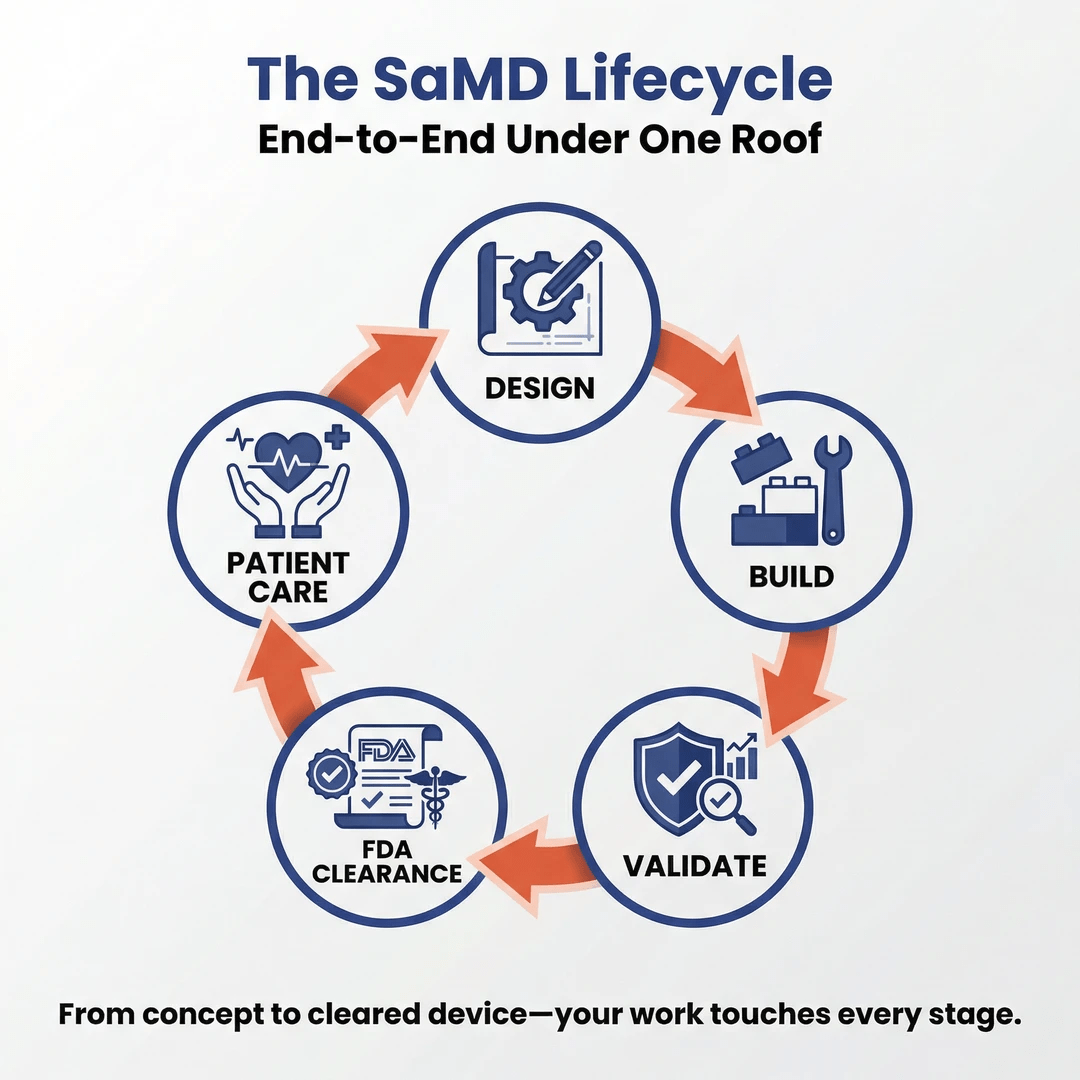
Innolitics is a specialized, remote-first firm focused exclusively on AI-enabled Software as a Medical Device (SaMD). We help visionary founders and companies design, build, validate, and obtain FDA clearance for clinical AI products—often cutting timelines from years to months. With 60+ devices successfully cleared and deep expertise in medical imaging AI and clinical decision support, we deliver end-to-end solutions under one roof: engineering, biostatistics, regulatory strategy, and cybersecurity. Our mission is to accelerate safe, effective AI into patient care while building meaningful, flexible, and financially rewarding careers.
The Opportunity 🔗

We are looking for an MD/PhD who is equally excited by rigorous biostatistics, writing production-quality code, and translating clinical insight into real-world AI tools. You will directly shape an innovative AI-powered SaMD project focused on medical imaging analysis and clinical decision support. Your work will combine statistical rigor for FDA validation, custom data pipelines and analysis tools, and close collaboration with our software and regulatory teams to help our clients to bring high-impact diagnostics to market faster.
This is not a pure research or pure coding role—it’s the perfect intersection where your clinical training, PhD-level stats expertise, and software skills will have immediate, measurable patient impact.
Key Responsibilities 🔗
- Design and lead biostatistical plans for AI model development, performance evaluation, clinical validation studies (e.g., MRMC, standalone, prospective), and real-world evidence generation
- Build and maintain production-grade statistical software, data pipelines, analysis tools, and visualization dashboards (Python/R primary)
- Develop robust performance metrics, confidence intervals, and statistical documentation for FDA submissions (510(k), De Novo, PMA)
- Analyze large clinical imaging and patient datasets; implement model validation, bias assessment, and generalizability studies
- Collaborate cross-functionally with software engineers, regulatory specialists, and clinical teams to integrate stats into the full SaMD lifecycle
- Contribute to open-source tools or internal platforms that accelerate AI medical device development
- Stay current and help the team grow through our weekly “10x Time” learning sessions on biostats, AI/ML, and regulatory topics
Required Qualifications (must-haves) 🔗
- MD degree and PhD in Biostatistics, Biomedical Informatics, Bioinformatics, Statistics, Epidemiology, or a closely related quantitative field
- Strong hands-on software development experience (fluent in Python and/or R; Git, SQL, Docker, ML frameworks such as scikit-learn/PyTorch/TensorFlow, or pipeline tools)
- Demonstrated expertise in applied biostatistics for clinical research, diagnostic tests, or AI/ML validation (publications, consulting, or industry experience)
- Ability to bridge clinical medicine, rigorous statistics, and production software engineering
- Passion for regulatory science and delivering AI that improves real patient outcomes
- Excellent written and verbal communication (you will write FDA-facing statistical sections and present to non-technical and FDA audiences)
- U.S. work authorization and location in continental United States
Preferred Qualifications 🔗
- Prior experience with medical imaging data (DICOM), genomics, EHR, or large multi-center clinical datasets
- Exposure to FDA medical device regulations or clinical trial design for diagnostics/AI
- Track record of building statistical software tools, GitHub contributions, or open-source packages
- Board certification or active clinical practice not required
What We Offer 🔗
- Competitive compensation package with performance bonuses and growth participation opportunities
- Full benefits package: health insurance, 401(k) with 3% company contribution, generous PTO (2–4 weeks), internet & equipment stipends
- True flexibility: work the hours that suit you (most ICs have <1 hour of meetings/day)
- High-impact, meaningful work on projects that reach real patients
- Small, elite team of MDs, PhDs, and world-class engineers who will help you level up
- Weekly dedicated learning time and opportunities to shape company direction
If you are an MD/PhD who codes, obsesses over statistical rigor, and wants your expertise to accelerate AI from bench to bedside via FDA-cleared products, we want to talk.
How to Apply 🔗

Send your CV, GitHub/profile link (or code samples), and a short video (up to 5 minutes) telling us:
- Why this role excites you
- One project or paper where your biostats + software skills made a difference
- A quick tour of your programming environment, including any AI assisted tooling you have used
How to Apply 🔗
Please email info@innolitics.com with the aforementioned informaiton. Add the name of this job ad in the subject (Physician-Scientist (MD/PhD) – Biostatistics & AI Software Development for Medical Device AI )
We are hiring as soon as we find the right person—so don’t hesitate to apply! If selected to move forward, you’ll hear from our team within 2–3 business days.



