Practical tips for medical-device software engineers, process, and tips

FeaturedArticleAI/ML
Fifty-one FDA-authorized pathology AI devices through April 2026, only seven of which are whole-slide imaging algorithms. This ...
 Yujan Shrestha
Yujan Shrestha

FeaturedArticleAI/ML
AI-assisted medical device development is FDA-compliant when done right. The obstacle isn’t regulation. It’s the messy codebase...
 Yujan Shrestha
Yujan Shrestha
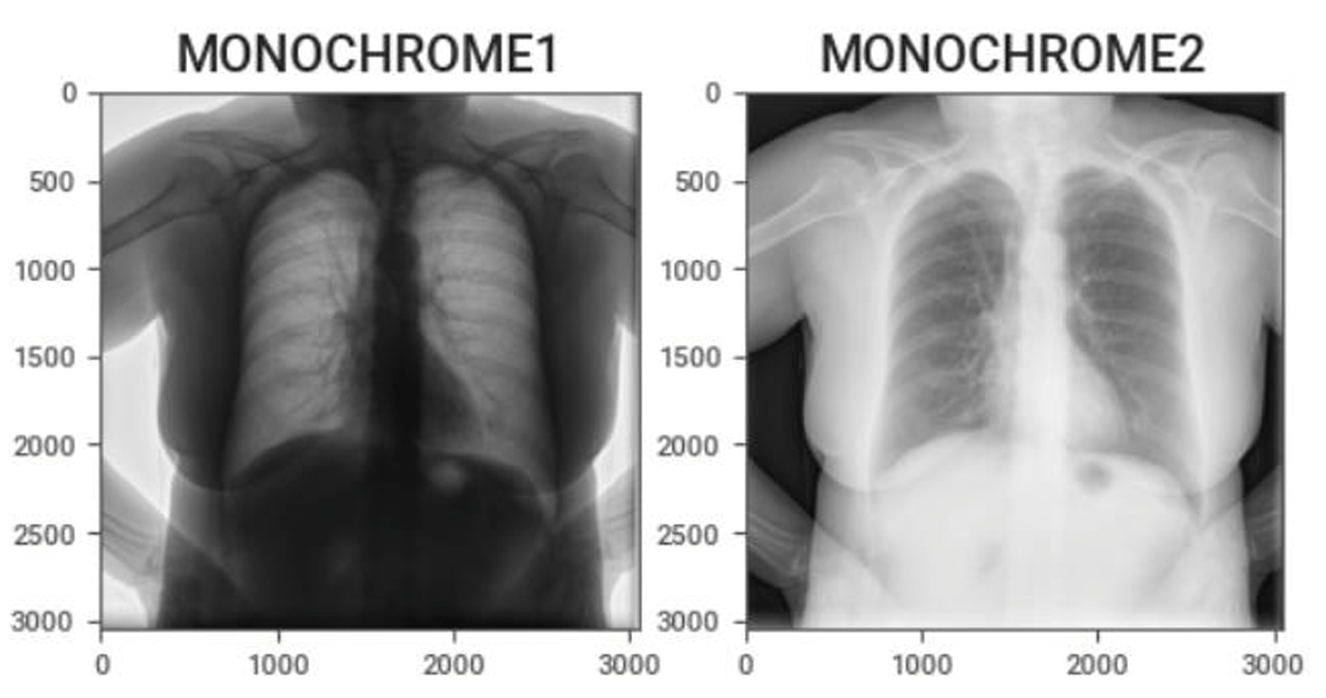
FeaturedArticleAI/ML
A practical guide for engineers new to medical imaging AI, highlighting common pitfalls—like coordinate-system errors and orien...
 Yujan Shrestha
Yujan Shrestha
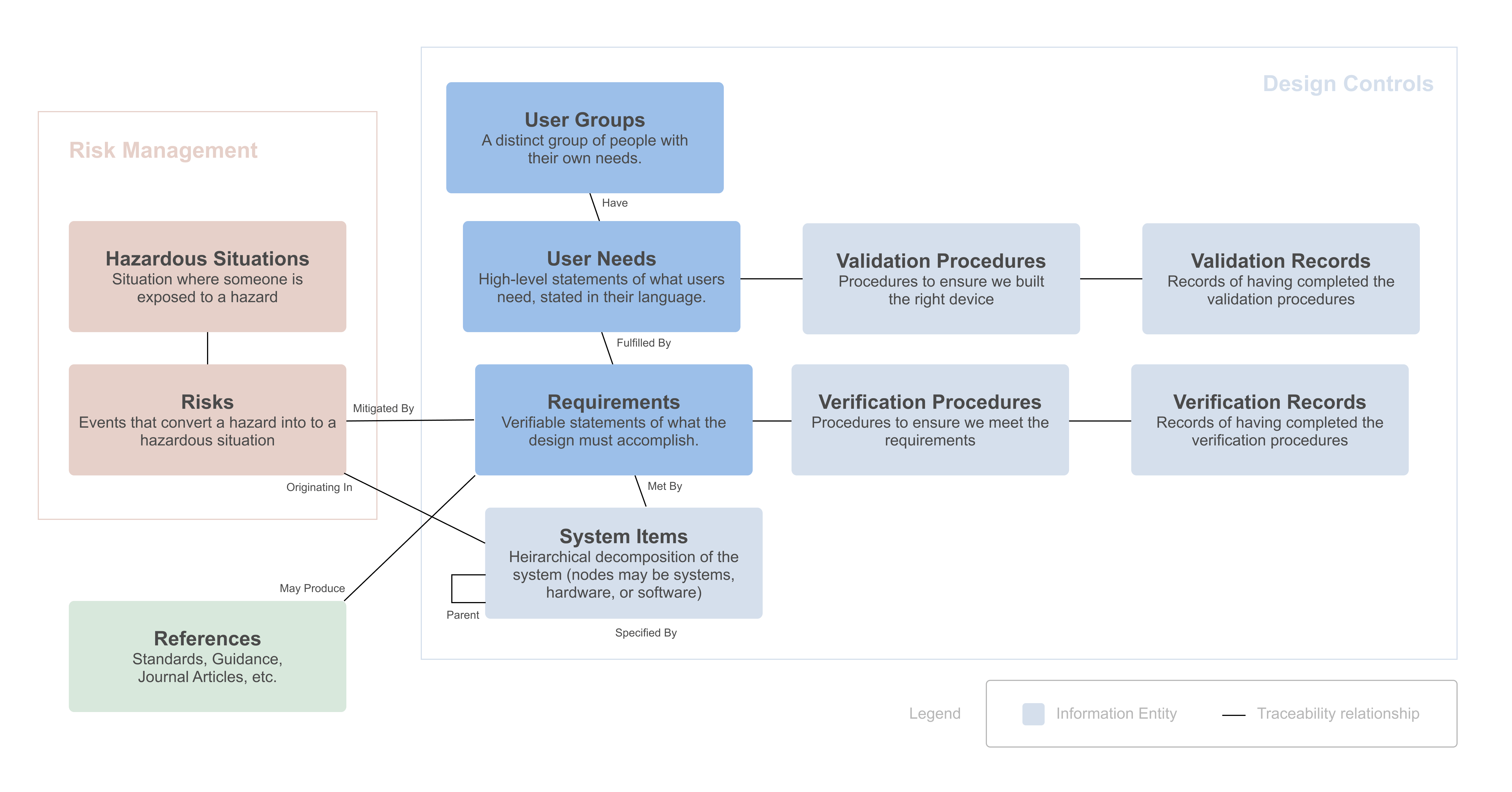
FeaturedArticleRegulatory
The FDA says developing your design inputs is “the single most important design control activity,” yet writing good design inpu...
 J. David Giese
J. David Giese
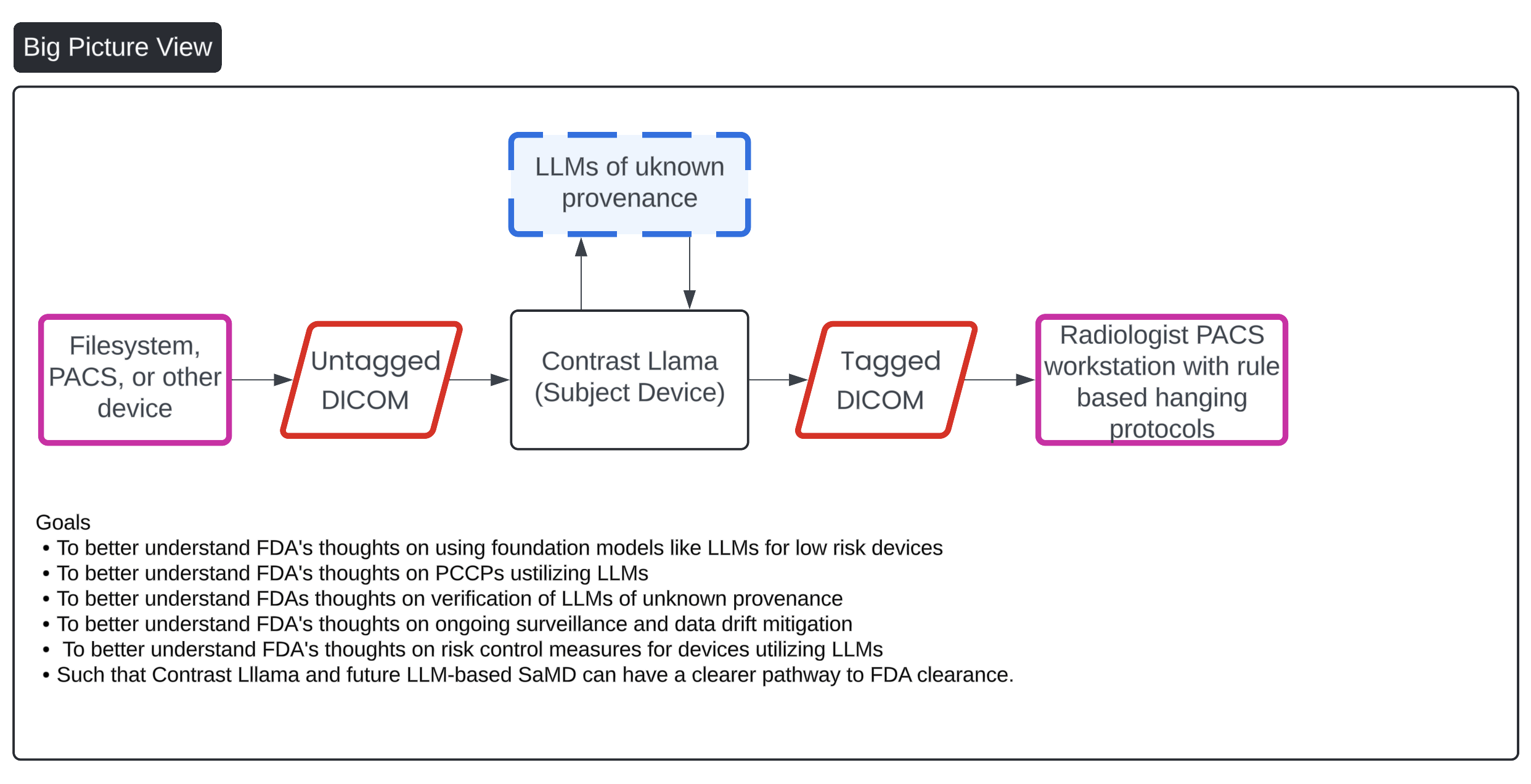
FeaturedArticleAI/ML
Ideas on how to get foundation models and generative AI FDA cleared.
 Yujan Shrestha
Yujan Shrestha

ArticleAI/ML
TytoCare's De Novo cracked open ENT, a specialty with just five prior AI/ML clearances, and dropped a 510(k) on-ramp for everyo...
 Yujan Shrestha
Yujan Shrestha

ArticleAI/ML
From solo clinicians prototyping algorithms to enterprise teams shipping production software, tools like Cursor, Claude Code, C...
 Yujan Shrestha
Yujan Shrestha
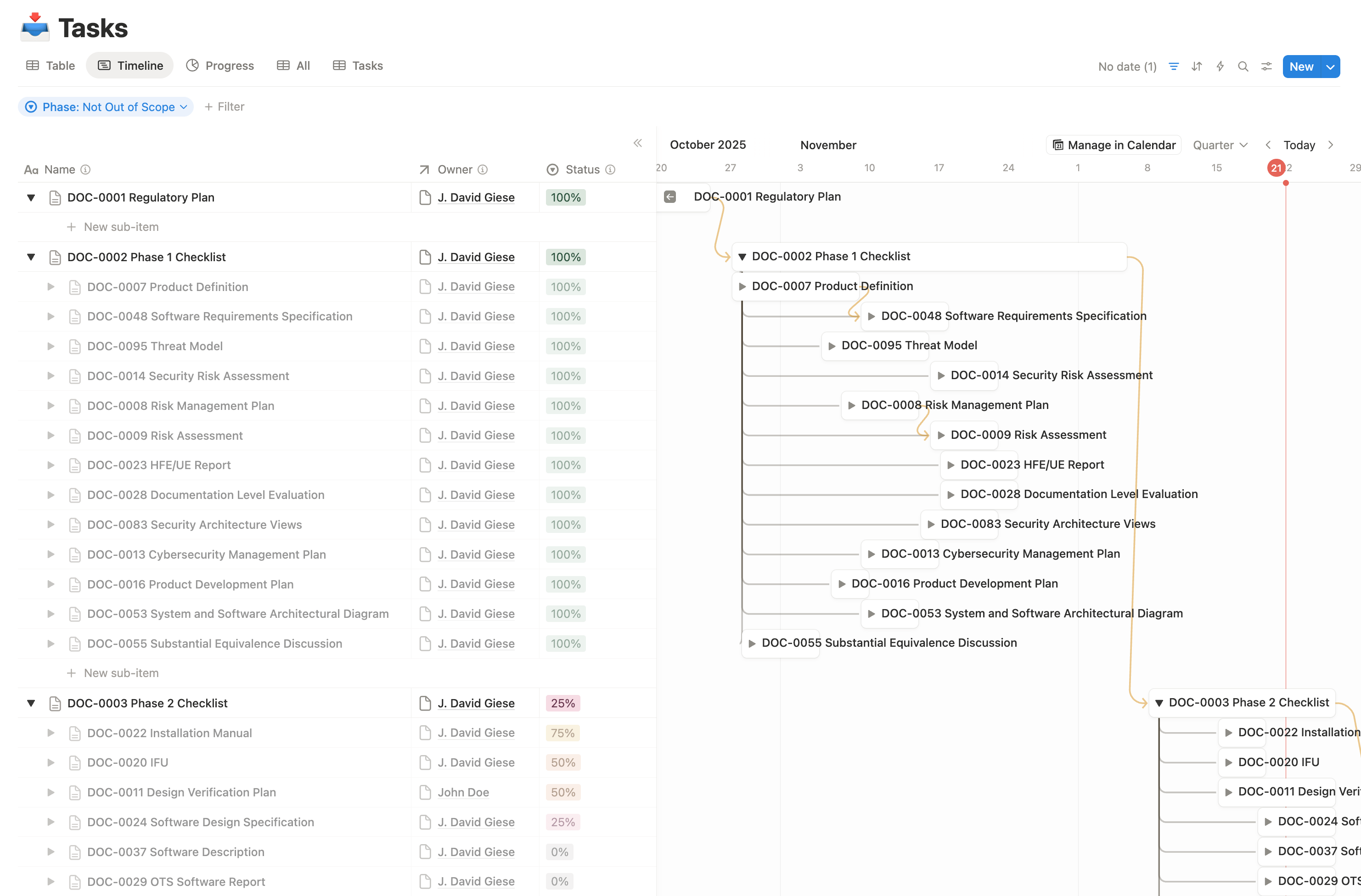
ArticleRegulatory
Are your medical device timelines slipping despite a smart team and lots of effort? This article explains Innolitics’ practical...
 J. David Giese
J. David Giese
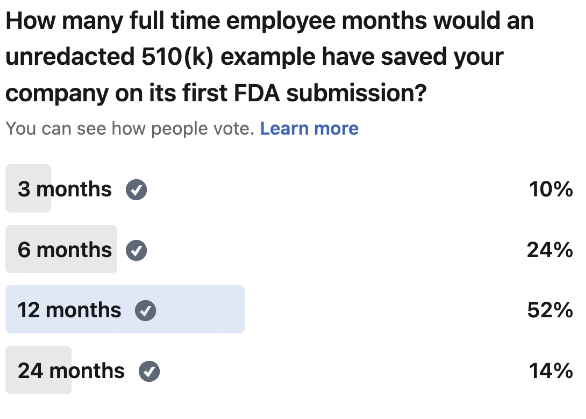
ArticleAI/ML
Most first-time teams burn over a year overbuilding documentation FDA never asks for. Here is a section-by-section breakdown of...
 Yujan Shrestha
Yujan Shrestha

ArticleRegulatory
To provide quality service to our clients, we need to do more than produce quality work.
 J. David Giese
J. David Giese
VideoRegulatory
The discussion in the transcript revolves around the FDA’s Draft Guidance on AI in Medical Devices, focusing on regulatory cons...
 Matt Hancock
Matt Hancock
VideoAI/ML
Kris Huang led a discussion on key image processing techniques in medical imaging, focusing on CT and MRI scans. Topics include...
 Kris Huang
Kris Huang
VideoAI/ML
Discussion covered key takeaways from the RSNA conference, balancing safety with speed, and setting risk thresholds. Topics inc...
 Yujan Shrestha & 2 others
Yujan Shrestha & 2 others
VideoSoftware
The discussion outlines the evolution of FDA regulations, common violations, and the enforcement structure. It emphasizes the i...
 George Hattub
George Hattub
VideoAI/ML
The discussion explores generative AI (GenAI) in medical devices, focusing on FDA's considerations for regulation, risks, and t...
 Yujan Shrestha & J. David Giese
Yujan Shrestha & J. David Giese
VideoCybersecurity
We discuss challenges in medical device cybersecurity, including balancing FDA guidelines with practical hospital and manufactu...
 Yujan Shrestha & J. David Giese
Yujan Shrestha & J. David Giese
VideoAI/ML
Yujan and David discussed the topic of FDA's performance in regulating AI medical devices, focusing on the ideal regulatory sit...
 Yujan Shrestha & J. David Giese
Yujan Shrestha & J. David Giese

ArticleSoftware
Notes on medtech funding, covering accelerators and funding sources.
 J. David Giese
J. David Giese
VideoAI/ML
A 10x talk exploring the basics of prompt engineering, followed by an open-ended discussion.

ArticleDICOM
A practical guide for integrating software with VistA, the VA’s PACS. This guide is based on our experience working through the...

ArticleSoftware
Overview of SMART on FHIR and when it’s a good approach to integrating your software into the clinical workflow. It includes a ...

ArticleRegulatory
An in-depth guide to navigating the use of off-the-shelf software (OTS) in the medical device industry. OTS software is general...
 J. David Giese
J. David Giese

ArticleCybersecurity
A short article that attempts to explain why badly made diagrams will confuse FDA reviewers and slow down your submissions. Inc...
 J. David Giese
J. David Giese

ArticleAI/ML
What is the difference between user needs and requirements?
 Yujan Shrestha
Yujan Shrestha
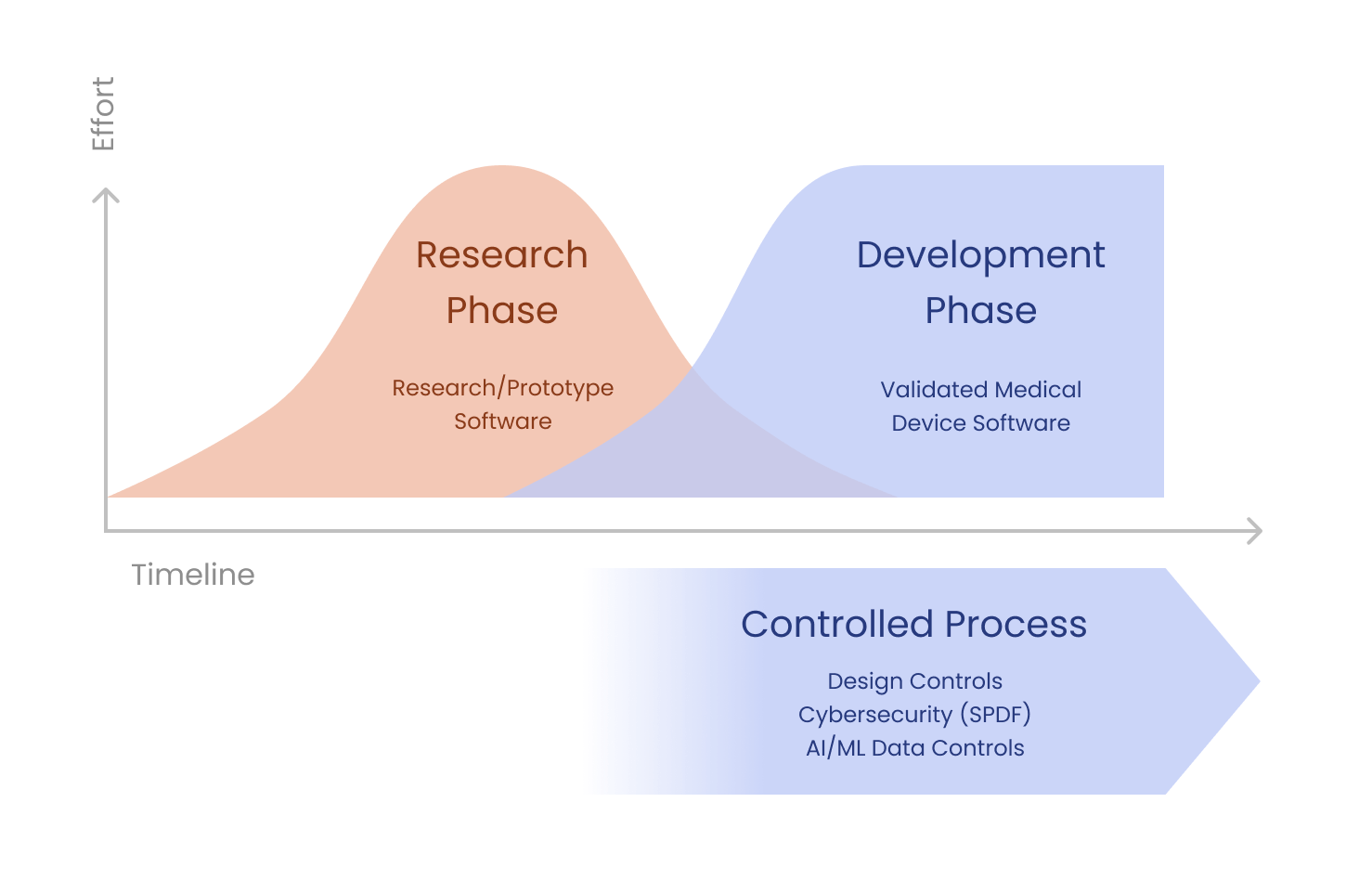
ArticleRegulatory
This article discusses engineering and regulatory factors to consider when deciding to rewrite or reuse existing prototype code...
 J. David Giese
J. David Giese
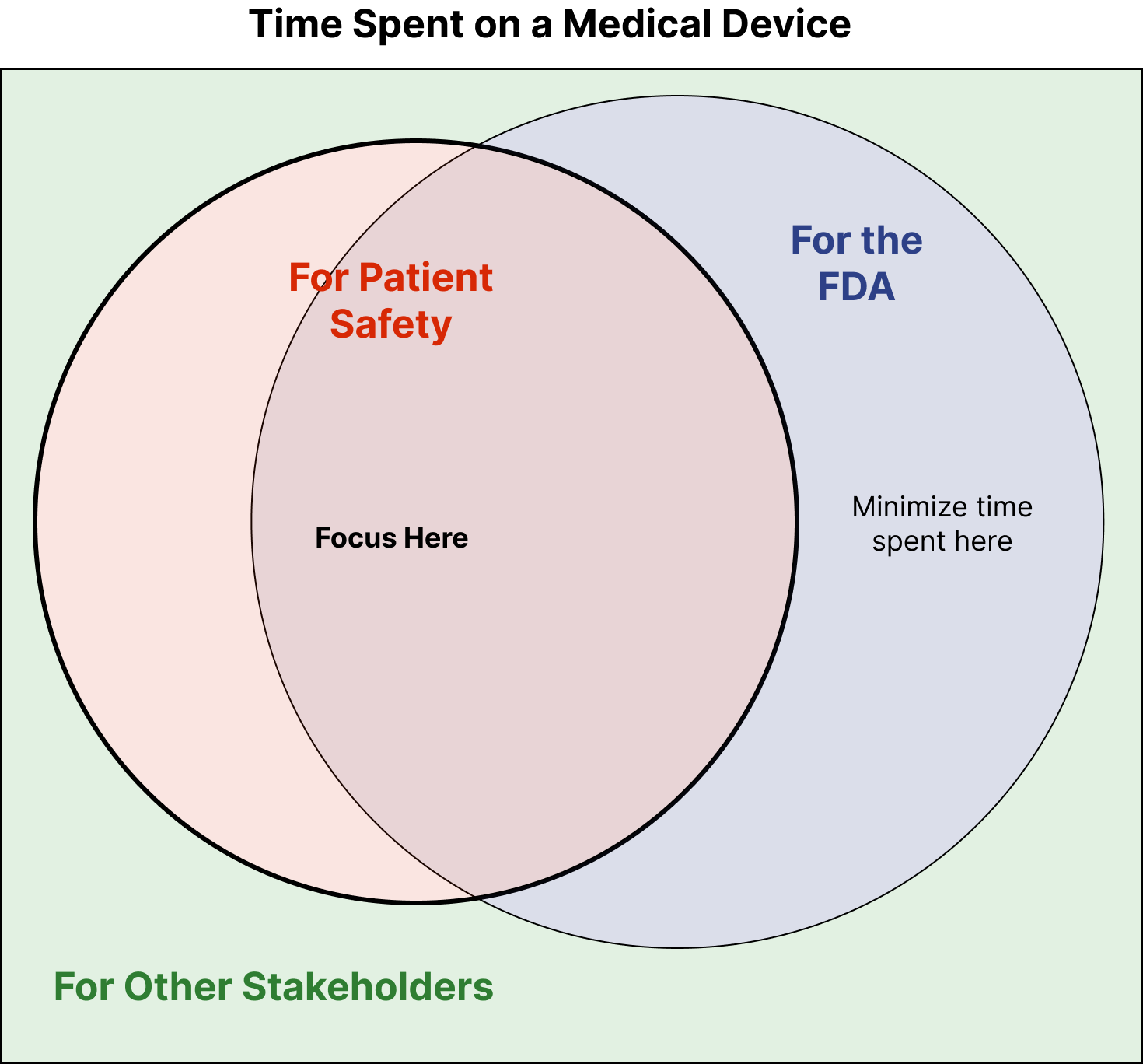
ArticleRegulatory
If you’ve ever been working on a medical-device and have thought, “this is a waste of time,” this article is for you. It will p...
 J. David Giese
J. David Giese
ArticleRegulatory
Medical-device software release frequency is a common question, particularly from software engineers familiar with agile develo...
 J. David Giese & Jim Luker
J. David Giese & Jim Luker
GuidanceRegulatory
This guidance document explains how medical device manufacturers can incorporate off-the-shelf software (OTS) into their device...

ArticleSoftware
This article explains why regulatory consultants who understand software can save your engineers time and help get your medical...
 J. David Giese
J. David Giese
VideoSoftware
Learn from a real-world MedTech OS software bug and explore various debugging techniques that can help you navigate through sof...
 J. David Giese
J. David Giese
VideoRegulatory
What is traceability? How does Notion help you handle it? How does Notion's flexibility empower medical device startups in ways...
 J. David Giese
J. David Giese
VideoSoftware
This video explains why code reviews are essential for growing a 10x software engineering team and how to do code reviews well.
 J. David Giese
J. David Giese
There are no resources matching these filter criteria.


Get To Market Faster
Our Medtech tips will help you get safe and effective Medtech software on the market faster. We cover regulatory process, AI/ML, software, cybersecurity, interoperability and more.