
REGULATORY AFFAIRS PROJECT MANAGER
Joined: February 13, 2023
I'm a biomedical engineer and regulatory professional. My journey began in a medical device startup, where I got to wear many hats. I decided to keep my favorite hat on, regulatory affairs, and my career has grown into that of a regulatory professional. Additionally, I'm passionate about guiding and mentoring young students interested in the field of biomedical engineering through my social media platform “The BME Life”.
YouTube, music, real estate, baking, and my two dalmatians.
Article
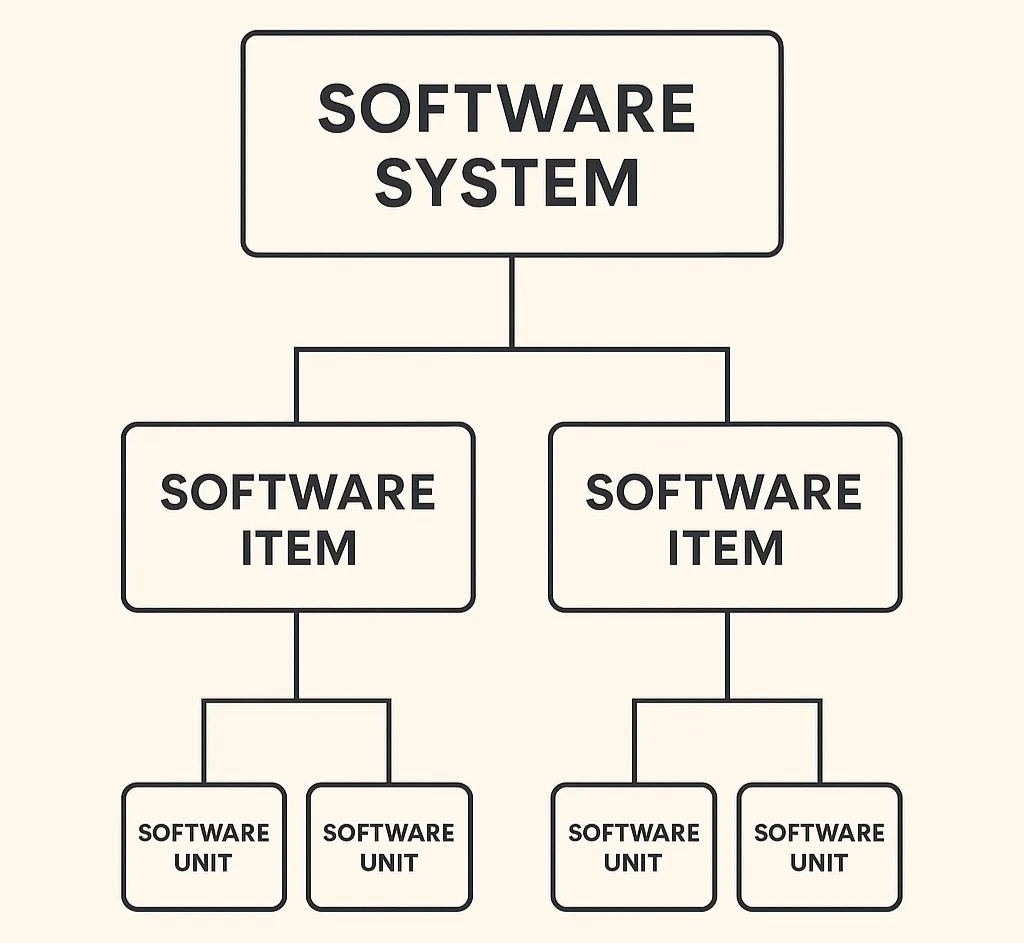
Practical tips on how to structure your SaMD architecture for FDA review without mirroring your codebase. Explains what the FDA...
 Meri Martinez & Yujan Shrestha
Meri Martinez & Yujan Shrestha
Article
A “quick” reference guide grouping the example 510(k) clearances by their product code/regulation, summarizing required testing...
 Meri Martinez
Meri Martinez

ArticleRegulatory
This checklist is meant to assist in the review of a 510(k) submission. It is a version of the checklist we use internally at I...
 Meri Martinez & 2 others
Meri Martinez & 2 others

ArticleRegulatory
This article provides an overview of FDA’s webinar regarding a set of guidance documents that were recently released by FDA as ...
 Meri Martinez
Meri Martinez

ArticleCybersecurity
This is a redline between the 2022 Draft cybersecurity guidance and the final 2023 cybersecurity guidance. It should be useful ...
 J. David Giese & Meri Martinez
J. David Giese & Meri Martinez