Medweb

CEO MedWeb
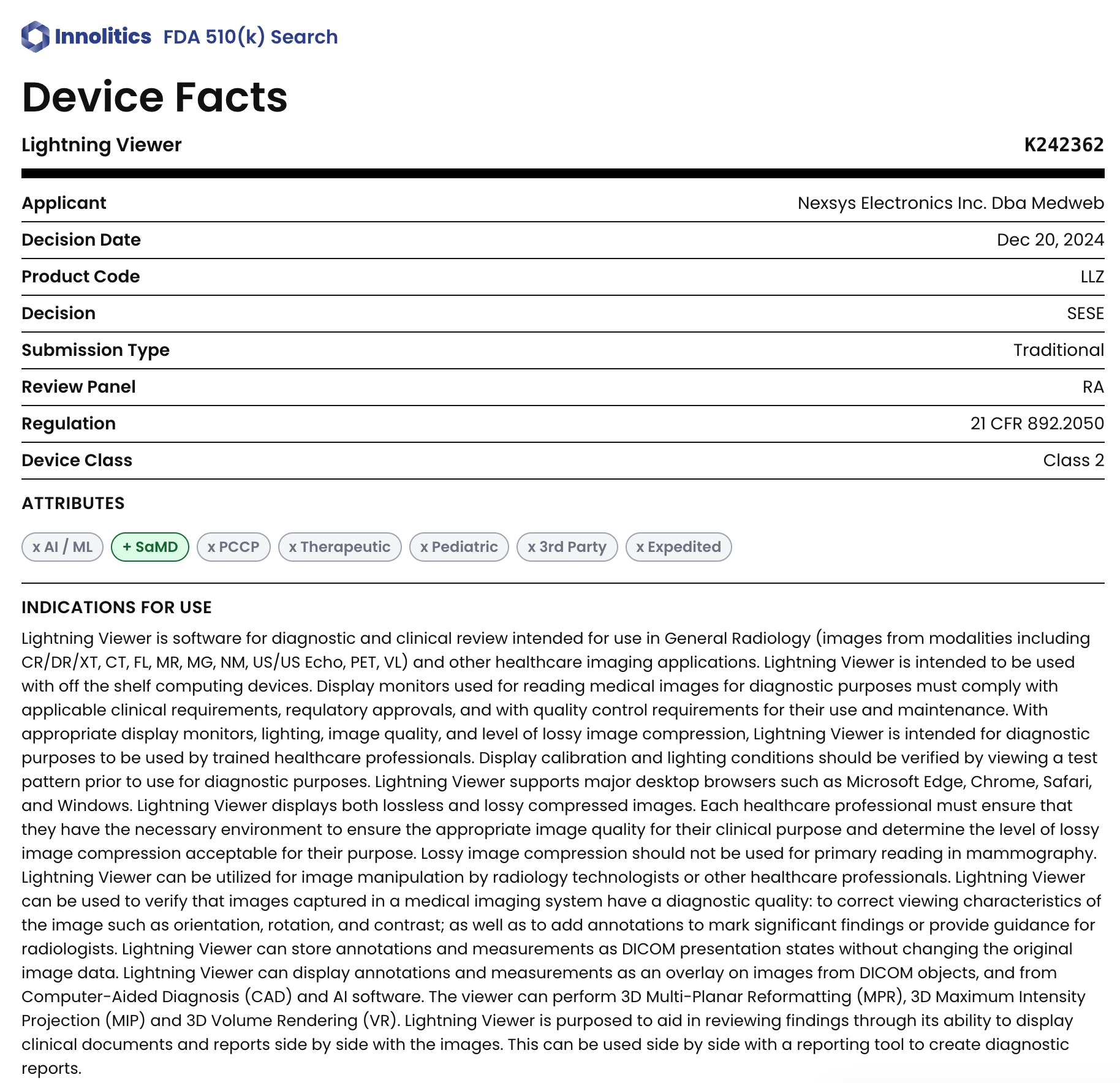
510(k) submitted in 7 weeks — one week ahead of an 8-week deadline. FDA clearance December 20, 2024, ~4 months after submission and 6 months after project kickoff.
Medweb came to us with a zero-footprint DICOM viewer and a hard deadline: clear the device in time for RSNA 2024. They needed a complete Design History File and 510(k) submission package built in eight weeks on top of engineering work that had never been packaged for FDA. No in-house regulatory writing capacity to hit that timeline alone.
Radiology
Prototype → 510(k) → QMS → FDA audit passed. Multi-year partnership with ongoing development.
Radiology
Gap analysis for an SaMD company that got audited that same year and passed with minimal findings
Radiology
Indistinguishable from Full-Time Staff. Software development and devops support for large existing engineering team
Every great partnership starts with a conversation. Fill out the form below for a discovery call, and an Innolitics team member will contact you soon.