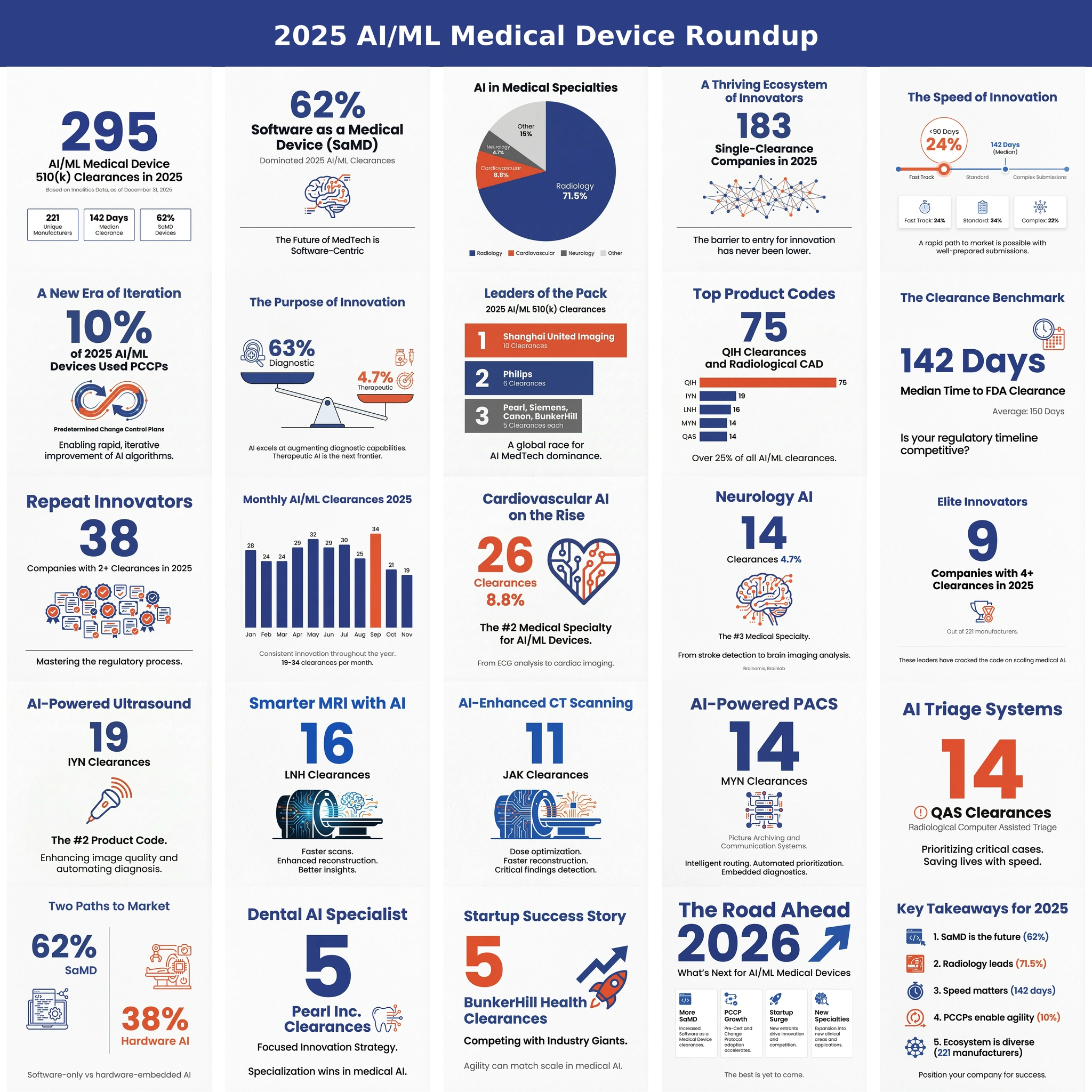
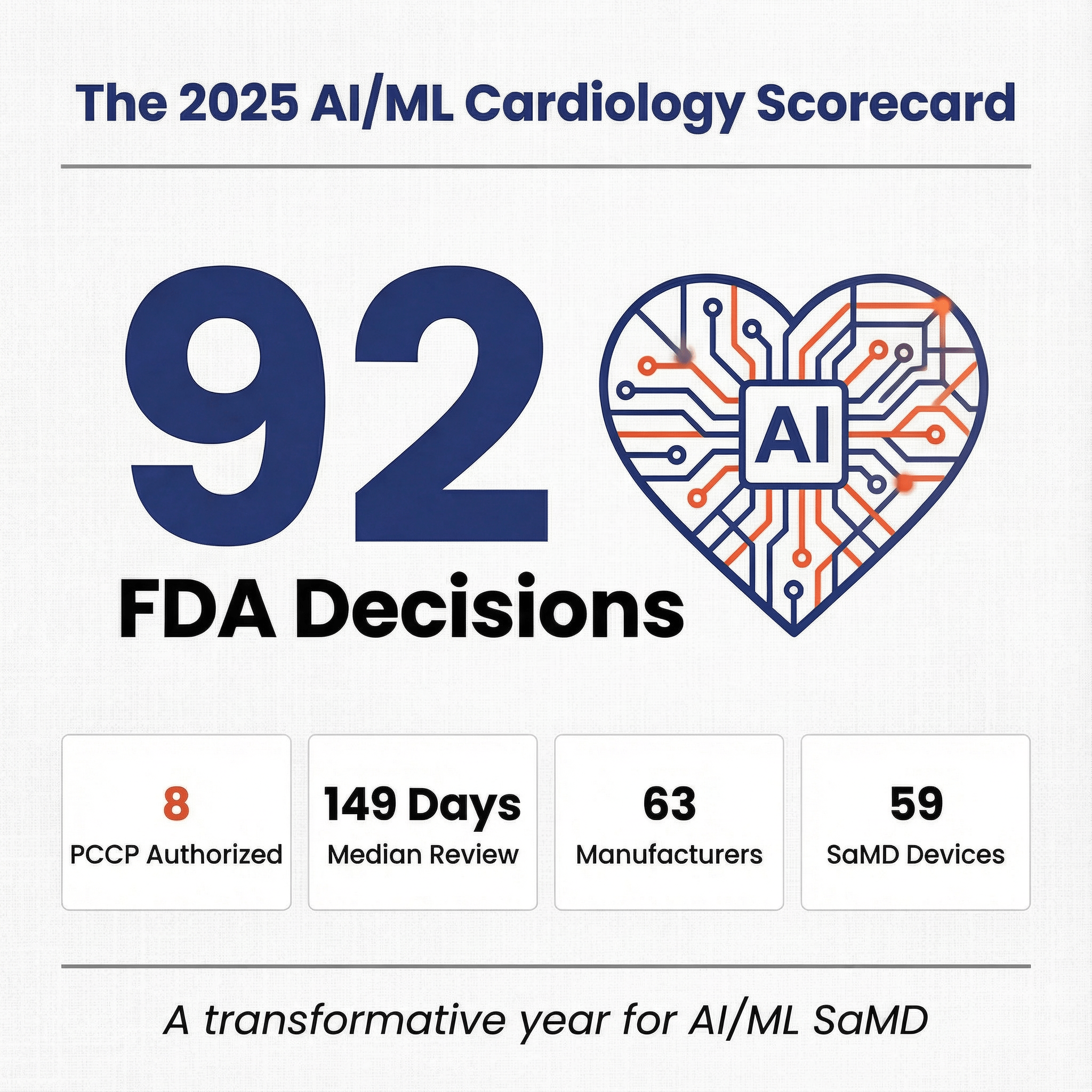
FDA cleared 24 unique AI/ML SaMD applications in March 2026. One every 31 hours. Three patterns worth mentioning: 79 percent landed in radiology, only one was a De Novo, and only two used a PCCP. Below, what those numbers tell you about the path of least resistance for SaMD in 2026.
Radiology dominates the pipeline 🔗
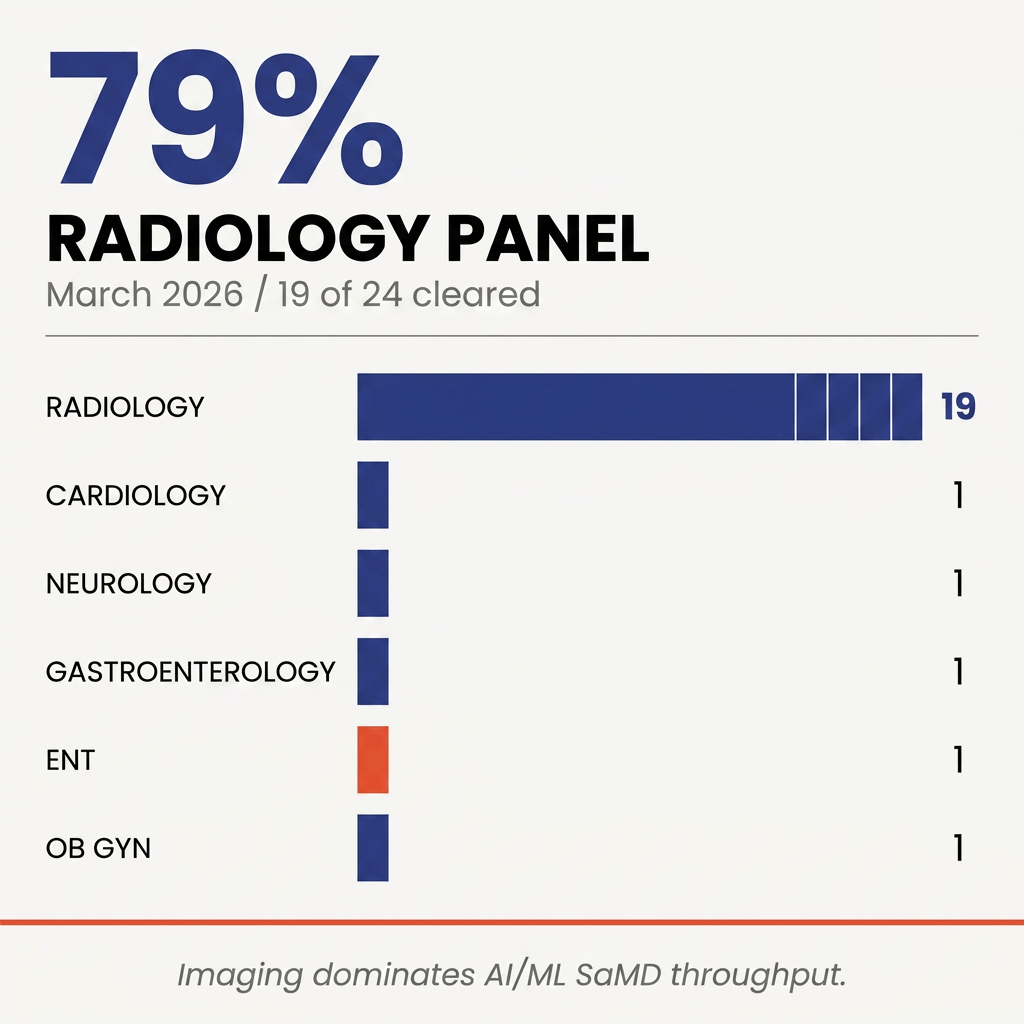
Nineteen of 24 clearances came through the Radiology panel. With a clean predicate and adequate validation, applicants clear in months, not years.
The breadth within radiology matters more than the share. Breast tomosynthesis (Lunit, K253796). Aortic valve calcium (BunkerHill, K260167). Acute stroke triage on CT (Harrison.ai, K253818). RT contouring (Varian K252304, Radformation K260509, MIM K253270). Surgical guidance (DePuy VELYS, K253551). Structural heart navigation (Philips EchoNavigator, K253614). All in one month.

Product code QIH alone covered seven of 24. QIH is anatomically agnostic: dental cephalometry, RECIST measurement, MR enhancement, and aortic planning all classify under it. For CADt submissions in 2026, QIH is the map most travelers use.
The De Novo standout: pediatric ENT 🔗

Tyto Care's Tyto Insights for Eardrum Bulging Detection (DEN250014) was the only De Novo in March. Cleared on first submission.
Pediatric otoscopy AI lacked an adequate predicate. ENT CAD has historically targeted adult endoscopy. A pediatric-specific eardrum tool occupies a regulatory gap.
Two takeaways. Sponsors targeting underserved subpopulations should not assume a predicate exists. De Novo may be the shortest path. And De Novo is not the harder path. Tyto cleared on first attempt. The FDA is making space for genuine novelty, not punishing it.
PCCP under-adoption 🔗
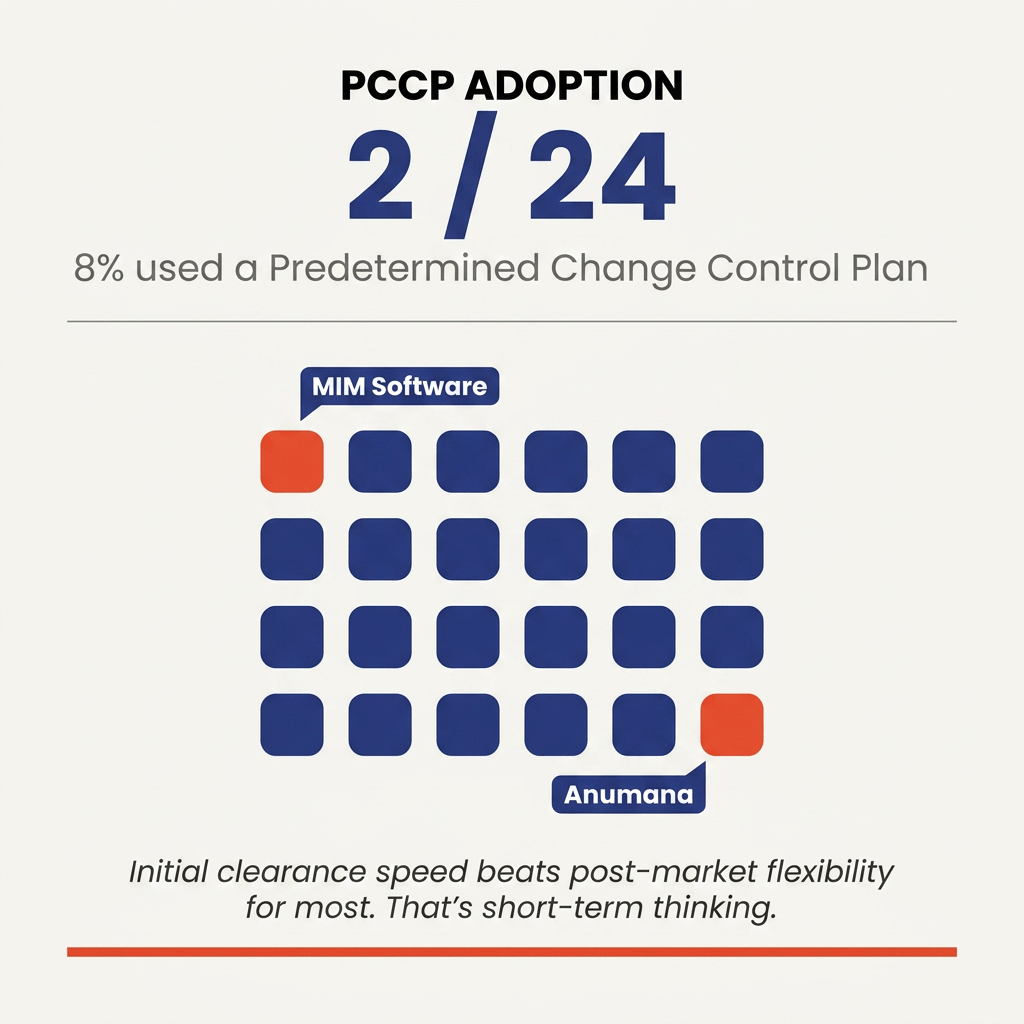
Only two of 24 applicants used a PCCP: MIM Software's Contour ProtegeAI+ (K253270) and Anumana's ECG-AI Pulmonary Hypertension tool (K252360).
PCCPs let sponsors modify a SaMD post-clearance without a new 510(k). FDA finalized the guidance in 2024. Eighteen months in, adoption sits at 8 percent.
Sponsors are optimizing for initial clearance speed, not post-market flexibility. That works for stable algorithms. It fails for any team planning frequent retraining. Without a PCCP, every meaningful update triggers a new 510(k).
If your algorithm will evolve, write the PCCP into the original submission. The companies that do this early compound their post-market advantage.
Geography: more than half from outside the US 🔗
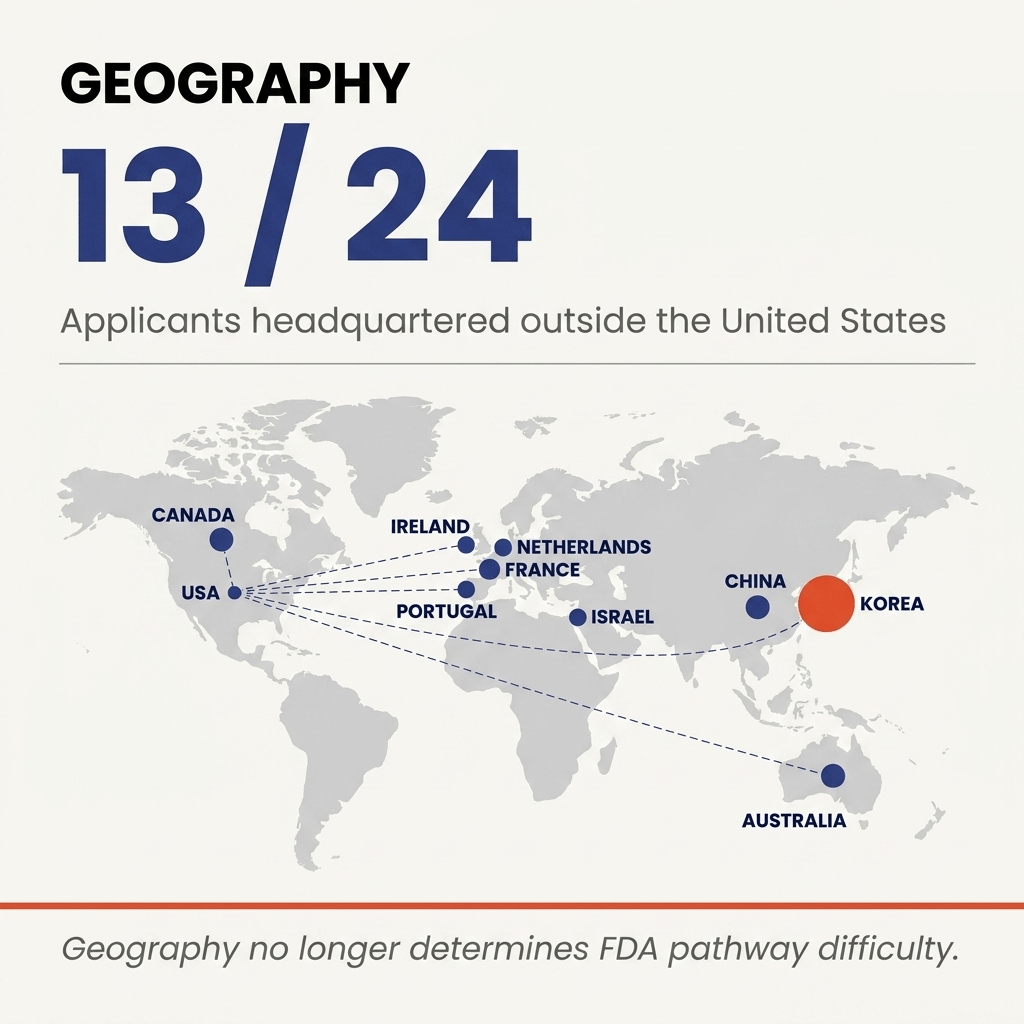
Thirteen of 24 applicants are headquartered outside the United States. Australia, Israel, South Korea, Netherlands, France, Portugal, Canada, China, Ireland.
Pediatric labeling stays scarce 🔗

Three of 24 March clearances carried pediatric labeling: Sonio Detect v3 (K252433), Tyto Insights (DEN250014), and SwiftMR (K253775). Pediatric AI is harder, and the gap does not close on its own. Sponsors who keep pediatric labels in their submission gain differentiated market position in an undersupplied niche.
Note: Sonio Detect v3 (K252433) is used for fetal ultrasound which technically is indicated for adult females but includes similar teaching points for multiple age subgroups.
How does this stack up against 2025? 🔗
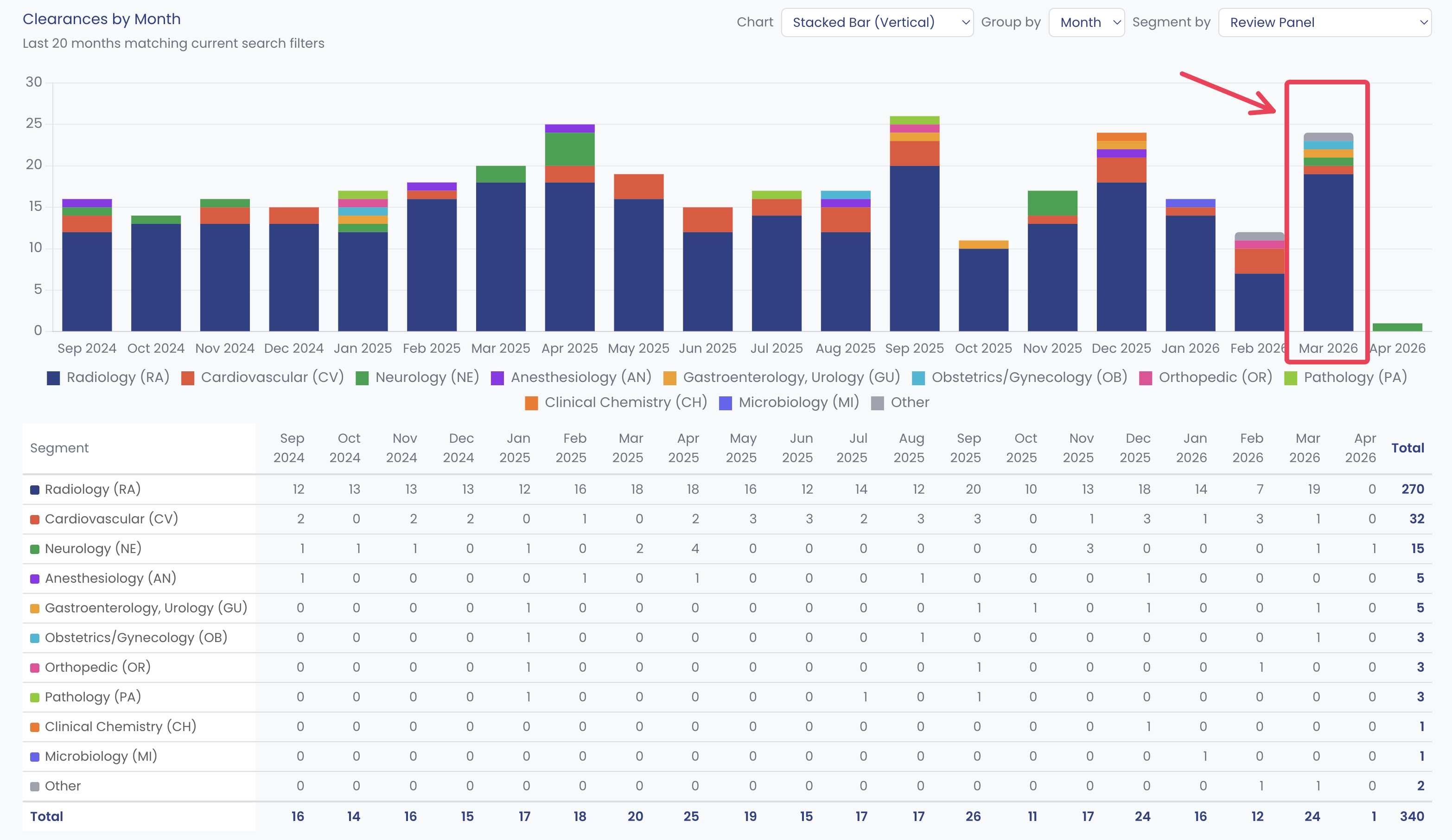
In our 2025 roundup, FDA cleared 295 AI/ML clearances, roughly 24.6 per month, with monthly totals ranging from 19 to 34. March 2025 cleared 20. March 2026 cleared 24. The pace is accelerating.
Takeaway 🔗
Radiology with a clean predicate is a months-long path. Identify the predicate first, validate against it, build the submission around that anchor.
Bake a PCCP into the original submission if your algorithm will evolve. Two of 24 did. The rest will fight individual 510(k)s for every update.
International sponsors cleared 13 of 24. Validation rigor and predicate clarity matter. Headquarters do not.
If you are working on therapeutic, closed-loop, or generative diagnostic AI, plan for longer timelines. The pathway is not blocked, but the validation playbooks are still consolidating. Engage the FDA early through a Q-submission before investing in large trials.
Twenty-four AI/ML SaMD applications cleared in March 2026. Nineteen in radiology with predicates in hand. One pediatric De Novo. Two PCCPs. The rest, efficient submissions anchored in clear predicates and validated algorithms. Not a bottleneck. A well-worn path that rewards predicate discipline.
Hire Innolitics to plan it, build it, or clear it 🔗
This is the landscape we work in every day. We have shipped FDA-cleared AI/ML SaMD and we hold ourselves accountable to outcomes most consultancies will not.
What we do for AI/ML SaMD teams targeting a 2026 submission:
- Predicate strategy. Map your indication to QIH, QAS, JAK, or the right adjacent code and lock the comparator before you spend pivotal-trial money. International HQ is not the barrier. Missing the predicate is.
- PCCP from day one. Define re-training, architecture, and input-expansion lanes up front, with verification protocols and lower-bound CI thresholds the FDA will accept. Ship model updates without re-submission.
- De Novo when it is faster. If you are targeting a novel device, we will tell you when De Novo is the shorter path and when it is not.
- Author the submission. 510(k)s, De Novos, and the MRMC, standalone, and human-factors studies behind them, sized to clear the lower-bound CI on your endpoint.
- Stand up the QMS. ISO 13485 / QMSR / IEC 62304-aligned, with the AI/ML-specific design controls layered on.
Innolitics guarantees 510(k) submission timeline when your algorithm is ready to containerize and we lead the submission. We guarantee 510(k) clearance when your algorithm passes the acceptance criteria we set during our strategy service.
If you are submitting an AI/ML SaMD in 2026, hire us to plan it, build it, or clear it.