On April 27, 2026, TytoCare announced that the FDA had granted a De Novo classification for Tyto Insights for Eardrum Bulging Detection, calling it the first AI-powered eardrum analysis software to clear the U.S. regulatory bar [1]. Reading just the press release, it would be easy to file the news away as "another AI clearance." Reading the FDA decision summary alongside the broader landscape of AI/ML in the ear, nose, and throat (ENT) panel tells a more interesting story: a quiet specialty has finally produced a regulatory template that a wave of follow-on devices can plug into [2] [3].
This article unpacks what the De Novo actually authorizes, why ENT has lagged the rest of medical-device AI, and what the submission reveals about how an over-the-counter diagnostic-aid device gets across the finish line in 2026.
A Blue Ocean Inside a Crowded Universe 🔗
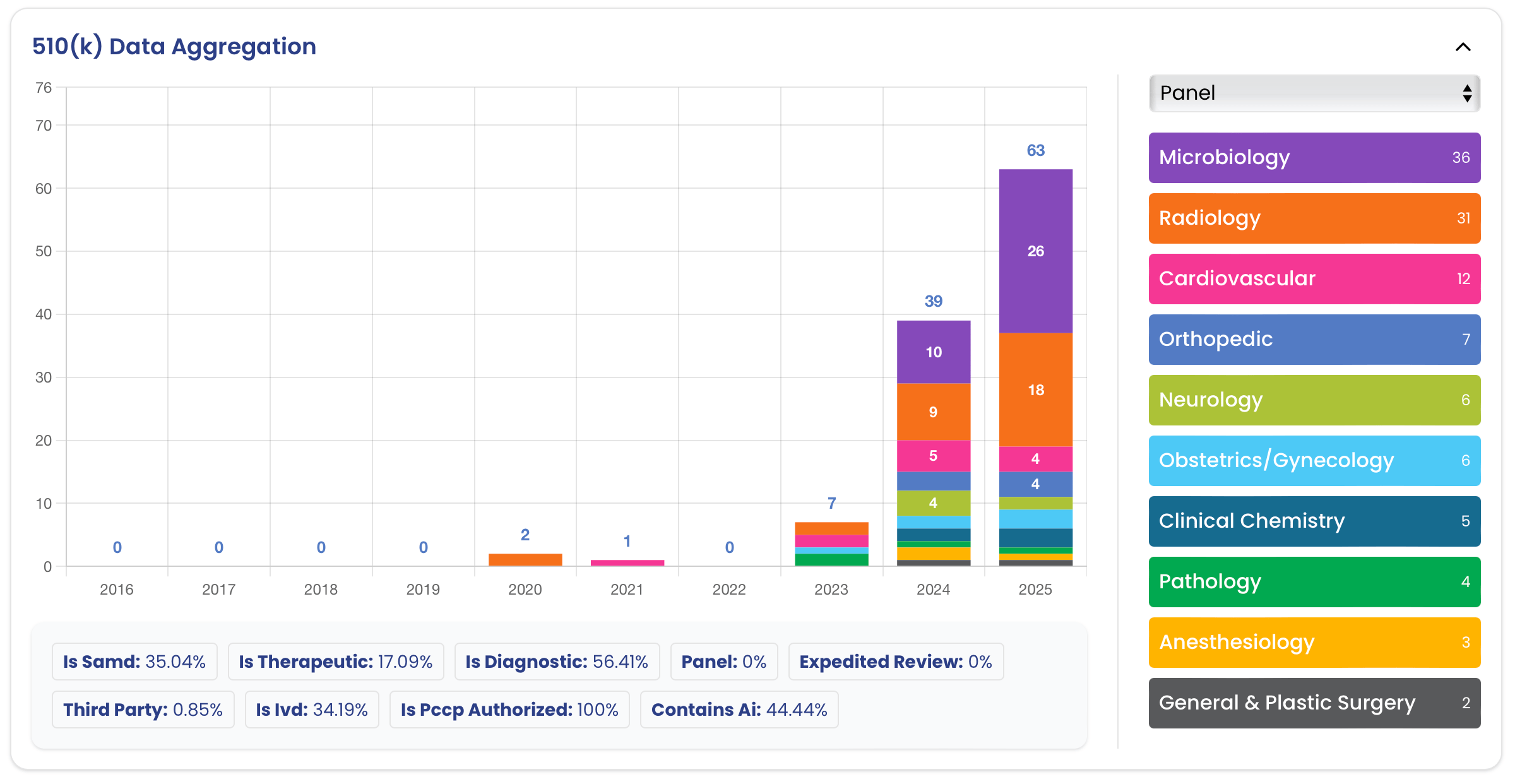
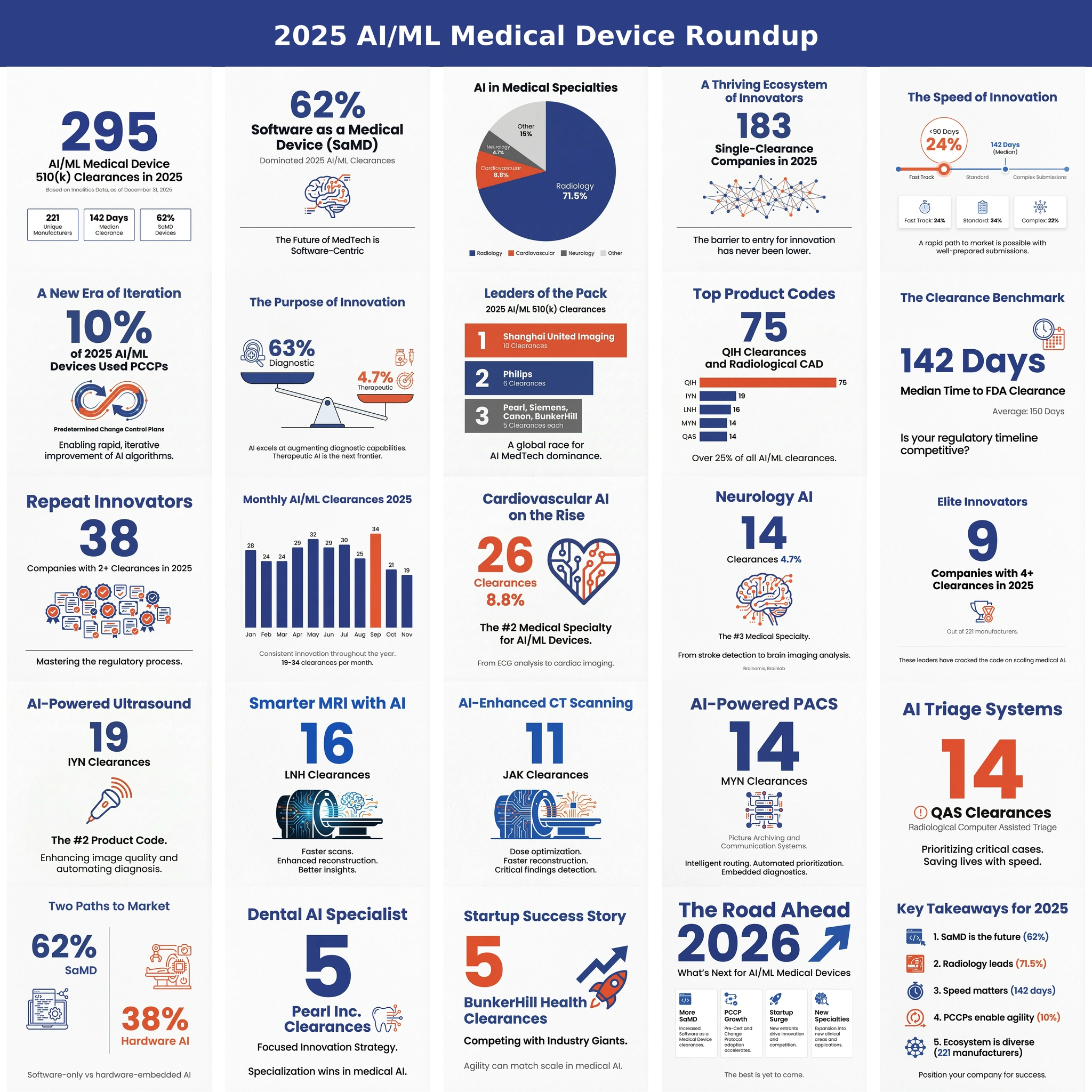
The FDA has now cleared or authorized roughly 1,569 devices that incorporate AI/ML across all clinical panels [3]. ENT is one of the smallest pockets of that universe. The ENT panel contains nearly 3,800 records spanning hearing aids, otoscopes, sinus stents, hearing-test software, vibratory therapies, and surgical tools. Of those, only five carry an AI/ML label: a hearing aid that self-fits, a stereotactic ENT navigation system, a software analyzer that estimates nasal congestion from imagery, an external mechanical sinus stimulator with adaptive software, and now Tyto Insights itself [3]. Two of the five are Software as a Medical Device, and only one is indicated for pediatric patients.

Put differently, ENT accounts for roughly 0.3 percent of historical AI/ML clearances despite representing nearly 3 percent of all FDA device records, an order of magnitude under-representation [3]. Several structural reasons explain the gap. ENT diagnostics have historically relied on subjective, qualitative assessments performed at arm's length with handheld instruments rather than on standardized digital images, so the kind of large, labeled datasets that radiology AI sits on simply did not exist for the middle ear or nasal cavity. Hearing-aid intelligence, where AI has quietly run for years inside on-device DSP, generally has not been positioned to FDA as a medical claim. And the most common ENT condition treated in primary care (acute otitis media) is diagnosed by a pediatrician or family-practice clinician with an otoscope in a few seconds, leaving little perceived headroom for software to add value.
That last assumption is precisely what TytoCare's submission challenges.
What the De Novo Actually Authorizes 🔗
The De Novo creates a brand-new generic device type called the "ear, nose, and throat image analyzer", codified at 21 CFR 874.4775, with a freshly minted product code (SHL) and Class II controls [2]. Tyto Insights is the first and currently only device in this class. Future ENT image-analysis software will now have a predicate to point at and a set of FDA-authored special controls to design against, which is the most consequential by-product of any De Novo: it converts an "uncharted" risk-classification problem into a 510(k) on-ramp for everyone who comes next.
The indication is narrow and carefully phrased. Tyto Insights is an over-the-counter, web-based AI software that automatically detects bulging of the eardrum in otoscopic video acquired by the compatible Tyto Otoscope, in patients aged six months and older, and is not intended to provide a diagnosis [2]. The labeling is required to remind users that a healthcare provider must interpret the result alongside the recording and the rest of the patient's clinical picture. The special controls themselves are noteworthy: clinical performance testing with explicit study endpoints and confidence intervals, human-factors testing for both clinicians and lay users, software verification, validation and hazard analysis, and labeling that includes hardware compatibility information and a clear "not-a-diagnostic" warning [2]. These are the rails any future SHL-coded device will run on.
The TytoCare De Novo was received on April 21, 2025 and granted on March 17, 2026, a 330-day review for a brand-new device type with a brand-new product code, an algorithmic engine, an OTC use case, and a pediatric population [2]. That is fast for a Direct De Novo of this complexity.
A Performance Story Built Against the Right Comparator 🔗
Most published AI imaging studies pit the algorithm against board-certified specialists. TytoCare's pivotal study did the opposite, and that is the most strategically important methodological choice in the entire submission. The validation set comprised 857 otoscopic recordings from 857 patients, balanced almost evenly between bulging-positive (429) and bulging-negative (428) cases [2]. The reference standard was the consensus of three U.S. ENT specialists, but the head-to-head comparator was a panel of three U.S.-licensed non-ENT clinicians (two pediatricians and one general practitioner) who represent the population that actually examines the vast majority of ears in this country [2].

Against that intended-use comparator, the device achieved standalone sensitivity of 92.31 percent (95% CI 89.02–94.43) and specificity of 94.63 percent (95% CI 92.27–96.56) [2]. The clinician panel achieved sensitivity of 72.42 percent and specificity of 73.25 percent. Because the lower bound of both differences exceeded zero, the primary endpoint was met for both sensitivity and specificity simultaneously [2]. The negative predictive value, prevalence-adjusted, was 99.29 percent [2].
The clinical insight worth dwelling on is the choice of denominator. By measuring its AI against generalist clinicians rather than against ENTs, TytoCare quantified the gap that already exists in routine primary care, then showed that its software closes a meaningful portion of it. That framing matters because it ties the benefit-risk argument directly to the OTC and primary-care reality: the question is no longer "is the AI as good as an expert?" but "does the AI raise the floor of the average examination?" For an over-the-counter device used by parents and front-line providers, that is the right question.
The Quality-Filter Safety Net 🔗
A second engineering choice is, on close reading, just as important as the bulging classifier itself. Tyto Insights does not feed every uploaded video into the diagnostic model. A separate AI component, the Eardrum Quality Filter, sits in front of the bulging classifier and decides whether the recording actually contains a sufficiently visible eardrum [2]. Videos that are too short, corrupted, or that fail to show the tympanic membrane are rejected before any clinical inference is attempted. The filter itself was independently characterized at 97.54 percent sensitivity and 89.97 percent specificity, with the sensitivity ground truth set by ENT specialists and the specificity ground truth set by the device's own optimal-frame detection [2].
This two-stage architecture is doing real work. The most predictable failure mode of an OTC otoscopy device is not a misclassified eardrum; it is a user who points the otoscope at the wax of the ear canal, the side of the canal wall, or nothing at all. By gating the diagnostic model on a quality check that itself was characterized clinically, TytoCare folded a substantial chunk of the human-factors risk into the algorithm rather than pushing it onto the labeling. Designers of any future at-home imaging AI, whether for skin, eye, throat, or wound, should study this pattern. It is a clean, generalizable mitigation.
The submission also reports a 15-clinician simulated-use study across three scenarios, plus a 30-person lay-user study split between adults aged 18–50 and adults aged 50 or older, supporting the claim that both clinicians and lay users can correctly use the system from the labeling alone [2]. Together, these studies are how the FDA convinced itself that the OTC indication for patients as young as six months would not produce systematic harm in real homes.
A PCCP That Lets the Algorithm Keep Learning 🔗
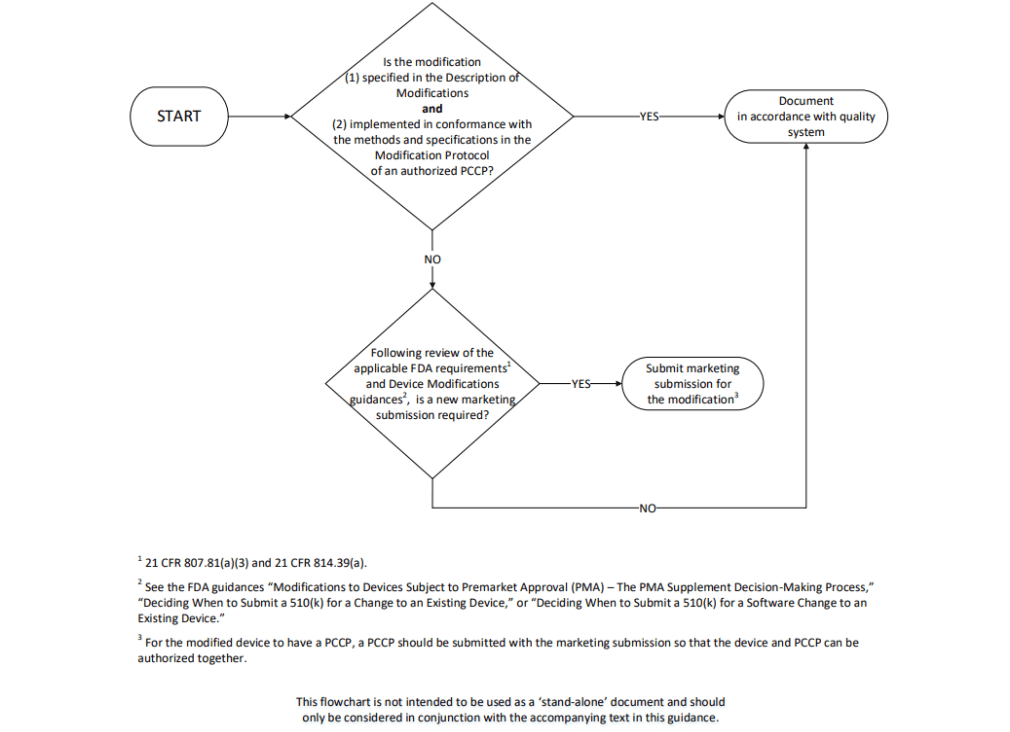
A forward-looking element of the authorization is the Predetermined Change Control Plan that the FDA approved alongside the device [2]. The PCCP defines, up front, three categories of post-market modifications TytoCare can ship without filing a new submission, provided each modification clears a pre-agreed verification protocol and continues to meet pre-specified performance goals defined by the lower bound of the two-sided 95% confidence intervals against the most recently validated release [2].

The first category is re-training: updating the bulging-detection model with additional otoscopic recordings to improve performance. The second is technical enhancement: changes to data preprocessing, data augmentation, model architecture, or hyperparameters intended to improve performance or computational efficiency. The third, narrower category is input expansion: adding compatibility for new versions of the Tyto Otoscope that meet predefined acquisition specifications: MP4 or comparable format, resolution at or above 640×480, field of view at least 45 degrees diagonal, and frame rate at least 10 frames per second [2]. Notably, third-party otoscopes are explicitly excluded from the PCCP scope; widening the device to a new hardware family would still require a separate submission.
Across the entire FDA AI/ML universe, only about 70 records carry an authorized PCCP today [3], so seeing one attached to a brand-new product code is a strong signal of where the agency expects the future of AI/ML SaMD to operate. For TytoCare specifically, the PCCP turns its growing library of real-world otoscopy recordings, a database the company says now exceeds 1.6 million ear images and recordings [1], into a regulatory asset rather than a regulatory liability.
A Decade of Hardware, Then a Decade of Algorithms 🔗
The eardrum-bulging De Novo is not a one-off. It is the most recent step in a clearance sequence stretching back to 2016, and the cadence is itself the strategy.

TytoCare first cleared a Tyto Stethoscope in 2016, a consumer-grade OTC variant in 2018, and a thermometer in 2019 [3]. Those three submissions were unremarkable Class II hardware clearances with no AI claim. Then, beginning in 2023, the company shifted its center of gravity. A Lung Sounds Analyzer (K221614) was followed by AI-enabled wheeze detection (K232237), crackles detection (K240555), and rhonchi detection (K243567), and most recently a third-generation stethoscope (K252089) cleared just five days before the eardrum De Novo [3]. The same week, the visual modality entered the portfolio with DEN250014.
The pattern is clean: deploy passive sensors at scale, gather labeled data through normal clinical use, and then unlock value, signal-by-signal, with a sequence of narrowly scoped algorithm clearances. Each new analyzer is independently submittable, independently labeled, and independently revenue-generating, but every one of them depends on the same underlying hardware footprint. For AI medical-device founders, the takeaway is that the clearance you ship in year ten can quietly be the most important asset of year one.
What This Means for the Next ENT AI Submission 🔗
Three lessons sit on top of the rest. First, a new generic-device classification with special controls is now in place for ENT image analyzers under 21 CFR 874.4775, and any sponsor proposing similar functionality (bulging detection, perforation detection, effusion grading, polyp detection, vocal-cord lesion detection) can now likely plan a 510(k) against the SHL predicate rather than mounting another De Novo from scratch [2]. Second, the FDA has accepted a non-specialist clinician panel as the right reader comparator for an OTC, primary-care AI diagnostic aid, which both lowers the bar for showing benefit and raises the bar for clinical relevance. Third, the FDA has accepted a PCCP that explicitly contemplates ongoing re-training and architecture changes for an SaMD with a pediatric OTC indication. That precedent will travel.
The bigger arc is the one to watch: ENT, dermatology, ophthalmology, and primary-care imaging in general are filled with conditions that are diagnosed in seconds by a clinician who may or may not have the training to do it confidently. The Tyto Insights authorization is a working template for converting that bedside, qualitative judgment into a regulated, software-mediated, continuously improving second opinion. In a specialty that until last month had effectively zero AI/ML SaMD on the market, that template is worth more than any single device.
Hire Innolitics to Build It or Clear It (or Both) 🔗
This is the space we work in. We partnered with PhotoniCare on their AI-powered middle-ear imaging platform, navigating the same architectural and regulatory choices that separate a 330-day clearance from a stalled program.
What we do for AI imaging device teams:
- Map your indication to the SHL product code (or the right adjacent one), then author the De Novo or 510(k) and shepherd it through FDA review.
- Design the PCCP up front so re-training, architecture changes, and hardware version bumps ship without re-submission.
- Build the SaMD itself: two-stage quality filtering, model architecture, inference pipeline, DICOM and EHR integrations, and a validation harness that doubles as your regulatory test record.
- Plan and run the MRMC and standalone studies against the right comparator panel for your intended use, sized to clear the lower-bound CI on your endpoint.
- Author the cybersecurity, SBOM, software, and risk-management documentation that FDA reviewers actually want, the first time.
- Run the Pre-Sub to lock the study design, predicate strategy, and clearance pathway before you spend pivotal-trial money.
- Stand up post-market surveillance and PCCP execution so your real-world data becomes the next clearance, not the next deficiency letter.
If you are building, validating, or pursuing FDA clearance for an ENT, dermatology, ophthalmology, or primary-care imaging AI device, hire Innolitics to build it or to clear it (or both).
References 🔗
[1] TytoCare. TytoCare Secures FDA De Novo for First AI-Powered Eardrum Analysis. Press release, April 27, 2026. https://www.tytocare.com/news-and-press/tytocare-secures-fda-de-novo-for-first-ai-powered-eardrum-analysis/
[2] U.S. Food and Drug Administration. De Novo Classification Request for Tyto Insights for Eardrum Bulging Detection. DEN250014, granted March 17, 2026.
[3] FDA Device Explorer dataset, queried April 28, 2026 (panel = Ear, Nose, Throat; AI/ML flag).
[4] Innolitics. 2025 Year in Review: AI/ML Medical Device 510(k) Clearances. https://innolitics.com/articles/year-in-review-ai-ml-medical-device-k-clearances/