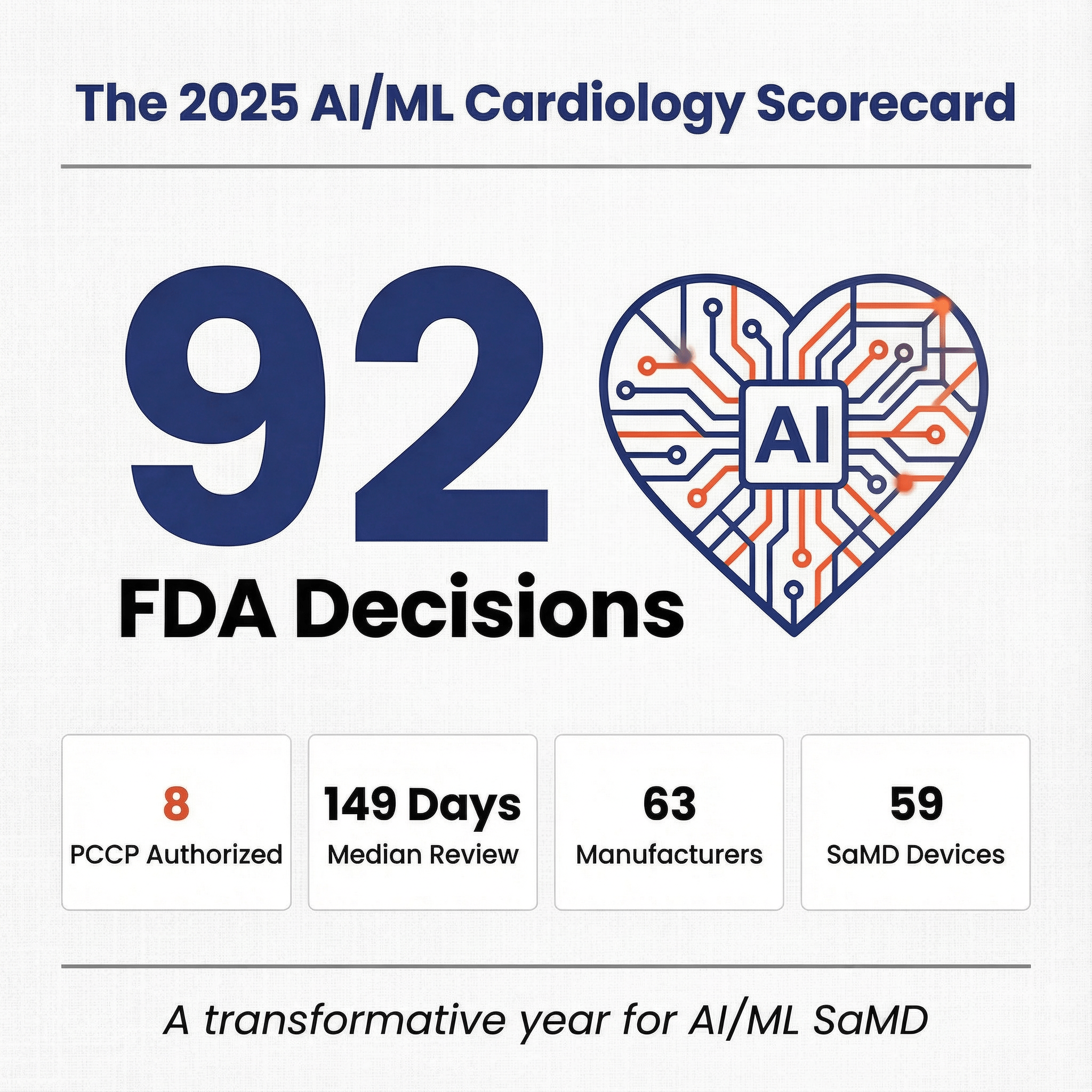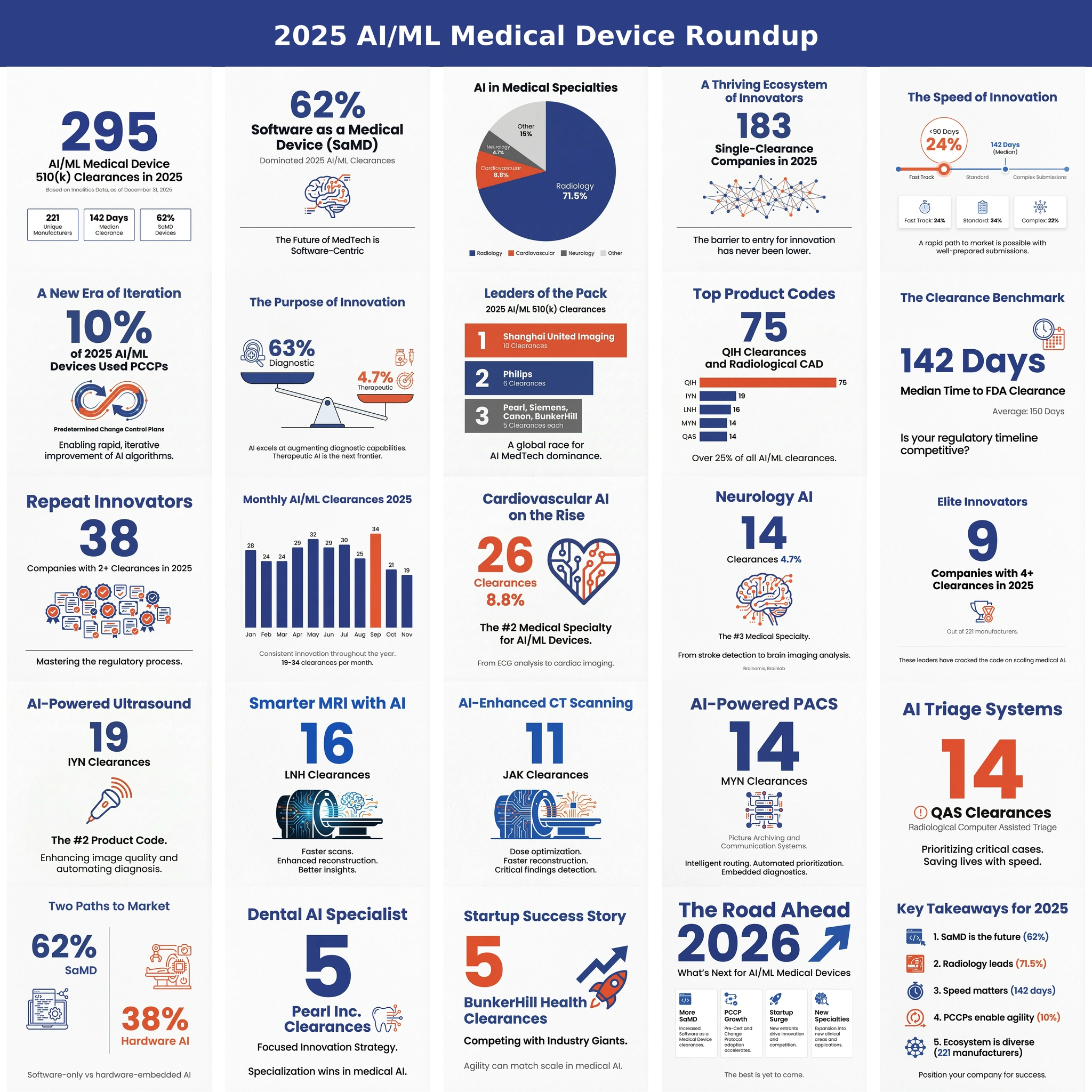
Discussions of clinical AI almost always start with radiology, for good reason. Roughly four out of five FDA-cleared AI/ML medical devices live in a radiology product code, the data fits cleanly into DICOM grids, and the workflow has clean handoffs between the modality, the PACS, and the reading room. Innolitics' 2025 Year-in-Review of AI/ML Medical Device 510(k) Clearances covered that story in detail. But while radiology dominates clearance volume, it does not dominate the most important structural change in the market.
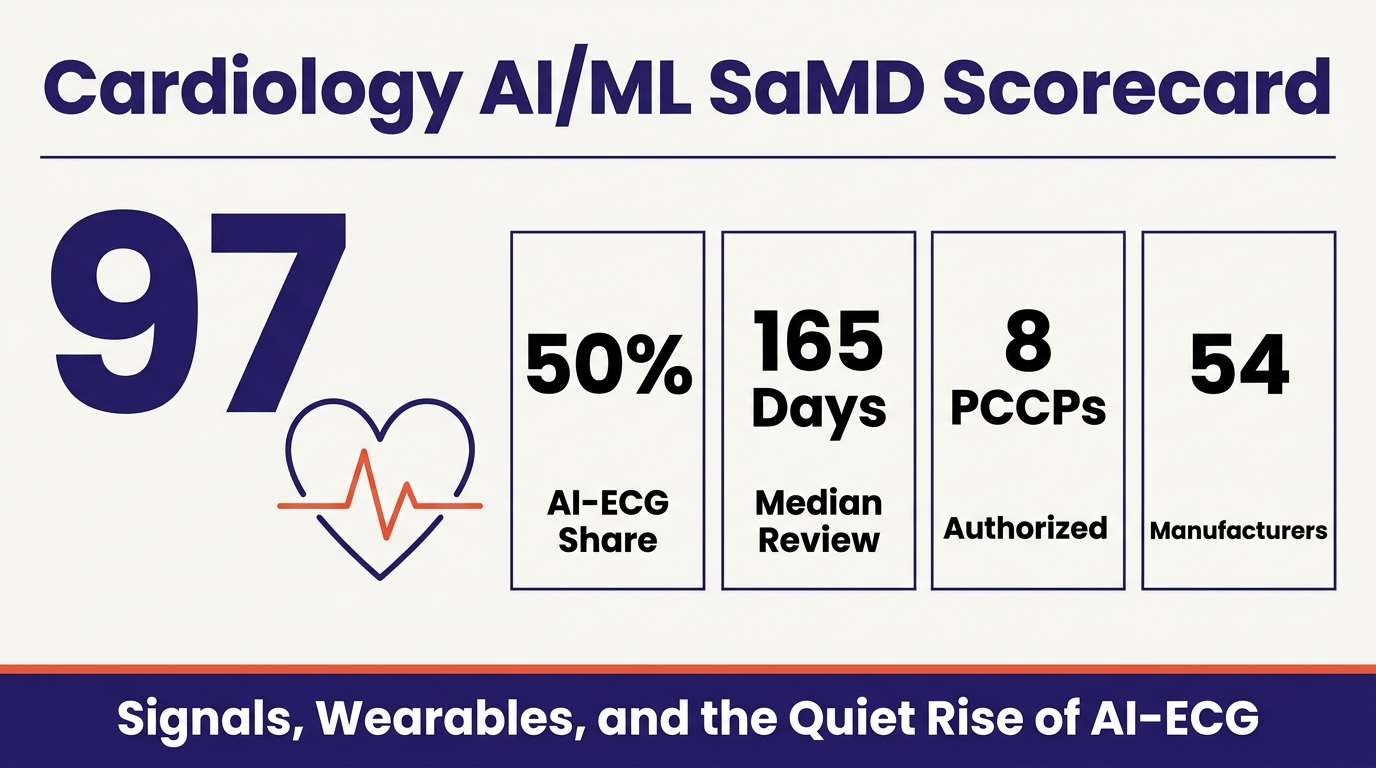
That change is happening in cardiology. Cardiovascular AI/ML SaMD is the second-largest specialty after radiology, with 97 cleared devices between 2006 and Q1 2026, per FDA Device Explorer data filtered to the Cardiovascular review panel, AI/ML usage, and SaMD classification. It is different from radiology in that it is a market for signals, not pixels: twelve-lead ECGs, single-lead patches, wrist PPG, chest phonocardiograms, and continuous arterial-pressure waveforms in the OR.
This article is a deep dive on that 97-device cohort, a companion to the radiology-heavy year-in-review using the same lens: what does FDA data tell us about how this part of the AI/ML SaMD market is actually being built?
How We Built the Cohort 🔗
The dataset behind this article comes from the FDA Device Explorer maintained by Innolitics. The exact filter set used to build the cohort is shown below.
| Filter | Value |
|---|---|
| Review panel | Cardiovascular (CV) |
| Contains AI/ML | True |
| Is SaMD | True |
| Decision date range | 2006-01-01 through 2026-03-31 |
The filter returns 97 records spanning twenty years and seven decision types, including Traditional 510(k), Special 510(k), and De Novo. None used the third-party 510(k) program or FDA's expedited/breakthrough pathway.
We then tagged every record using substring matches on cleared device name and intended-use text. Keywords like "ECG", "PPG", "echocardiograph", "Holter", "FFR", "plaque", and "amyloidosis" mapped each device to one or more of seven archetypes. Because a single device can satisfy multiple archetypes (e.g., a wearable PPG device that also detects atrial fibrillation), archetype counts sum to more than 97. Archetype shares are reported as a fraction of the 97 distinct devices.
The Shape of the Market: Twenty Years of Slow Growth, Then Three Years of Acceleration 🔗
The first decade of the dataset looks like a niche. Between 2006 and 2017, just six AI/ML SaMD devices cleared in the cardiovascular panel: Eko's auscultation analysis, an early Cardiologs Holter, the Hypertension Diagnostics DXH product, Biovitals' continuous patient analytics, and a few others. The cumulative total at the end of 2017 was seven.
The inflection point was 2018. That year produced nine clearances and three De Novo authorizations that created the modern regulatory architecture for cardiology AI. Apple secured a De Novo for the ECG App on Apple Watch (DEN180044), opening product code QDA: Electrocardiograph software for over-the-counter use. The same week, Apple secured a second De Novo for the Irregular Rhythm Notification Feature (DEN180042), opening QDB: Photoplethysmograph analysis software for over-the-counter use for opportunistic AFib detection. Earlier that year, Edwards Lifesciences secured DEN160044 for the Acumen Hypotension Prediction Index (HPI) Software Feature, opening QAQ for AI-based hemodynamic prediction in surgical and non-surgical patients.
These three De Novos did more than approve three products: they established three new categories of cardiac AI, consumer-grade single-lead ECG, opportunistic PPG-based arrhythmia screening, and AI-based predictive hemodynamics. Most cleared AI-ECG and AI-PPG devices since trace back to one of those three lineages.
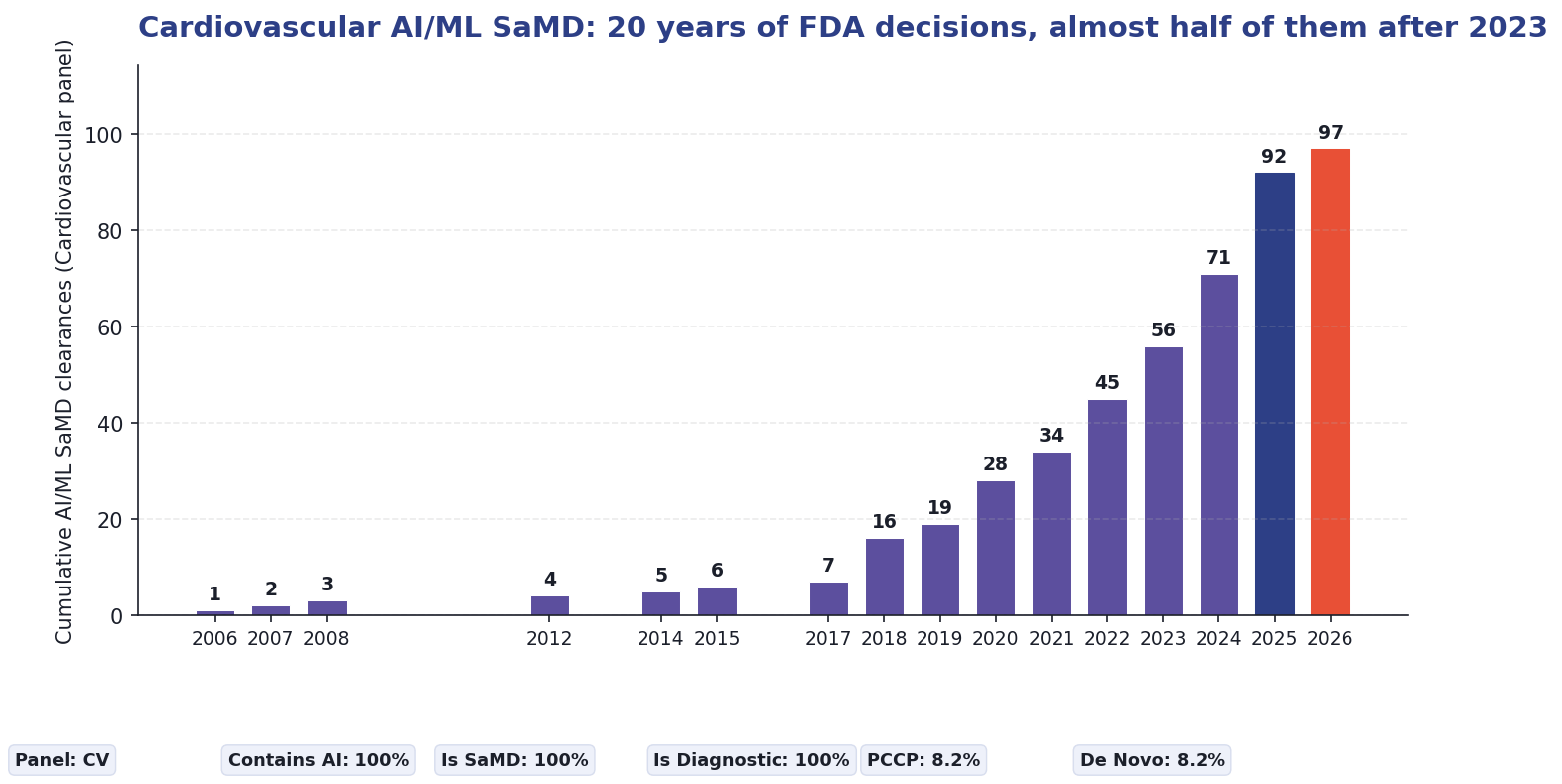
Since 2018, growth has compounded. The cumulative count passed 28 at the end of 2020, 56 at the end of 2023, and 92 at the end of 2025. Q1 2026 added five more, bringing the total to 97. More than a third of all cardiology AI/ML SaMD clearances in FDA history occurred in the last three years. The curve mirrors radiology AI in 2017–2020: rapid post-De Novo expansion as predicates accumulated and pathways became predictable. The radiology curve eventually flattened as the easy archetypes (mammography CAD, ICH triage, LVO detection) saturated. The cardiology curve has not yet reached that point.
The market is also less consolidated than it looks. The 97 devices span 54 manufacturers, but the distribution is lopsided: 34 of 54 (63%) have a single clearance, and only five hold four or more. The leaders mix consumer electronics, traditional medtech, and ambulatory-monitoring specialists.
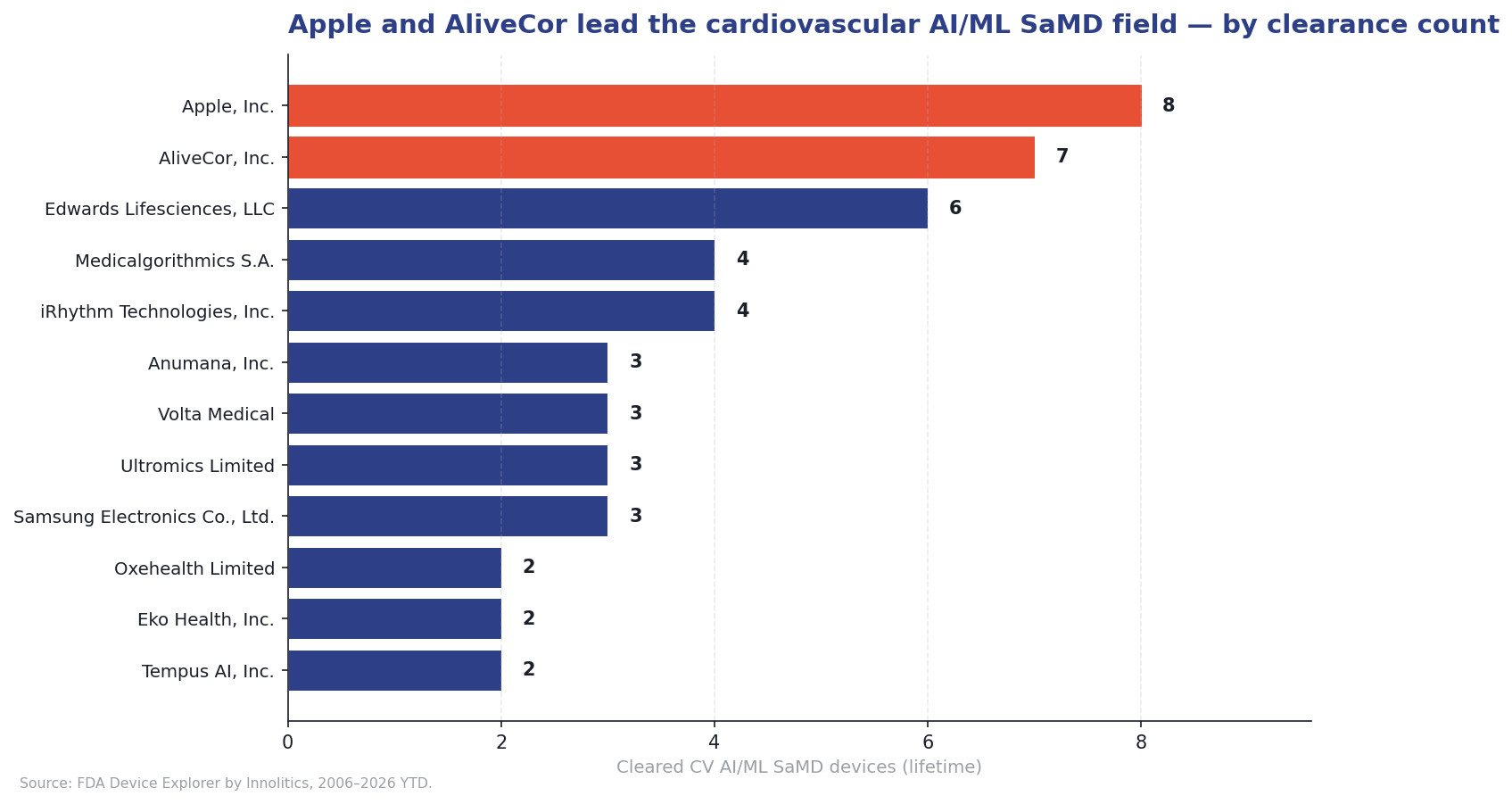
Apple leads with eight clearances, followed by AliveCor (seven), Edwards Lifesciences (six), and Medicalgorithmics and iRhythm tied at four. Anumana, Volta Medical, Ultromics, and Samsung are tied at three. Two consumer-electronics companies in the top group (Apple and Samsung) is unusual: no consumer-electronics company sits near the top of the radiology AI leaderboard. In cardiology they are setting the regulatory pace.
The Four Pillars of Cardiology AI 🔗
Radiology AI fits in one sentence: AI assists the radiologist in interpreting an image. The clinical surface area for Cardiology is broader and the data modalities more varied. Across the 97 devices, four primary archetypes emerge, with three smaller adjacents.
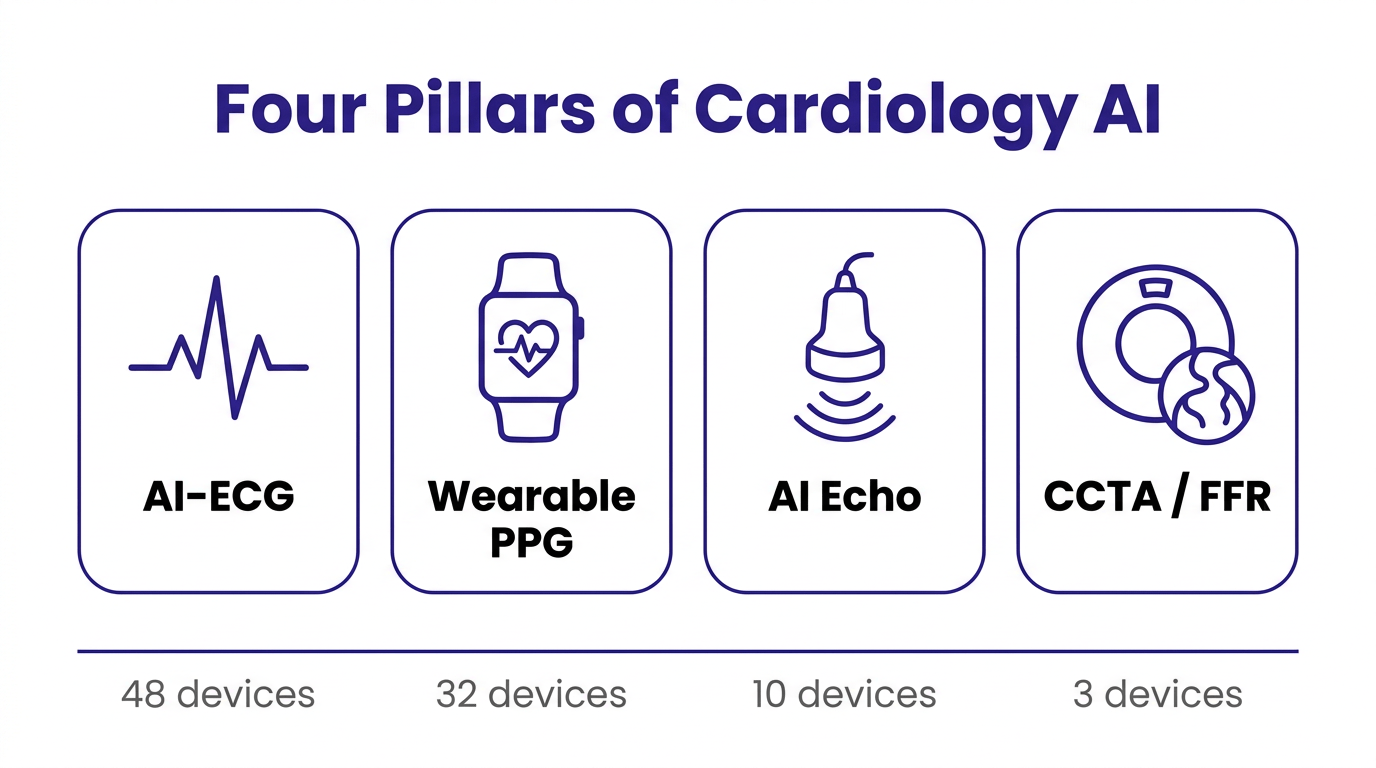
The four primary pillars are AI on the electrocardiogram (AI-ECG), AI on wearable PPG signals, AI on echocardiographic images, and AI on coronary CT angiography (CCTA) including FFR-CT. Each pillar has a different patient population, a different regulatory pattern, and a different commercial logic.
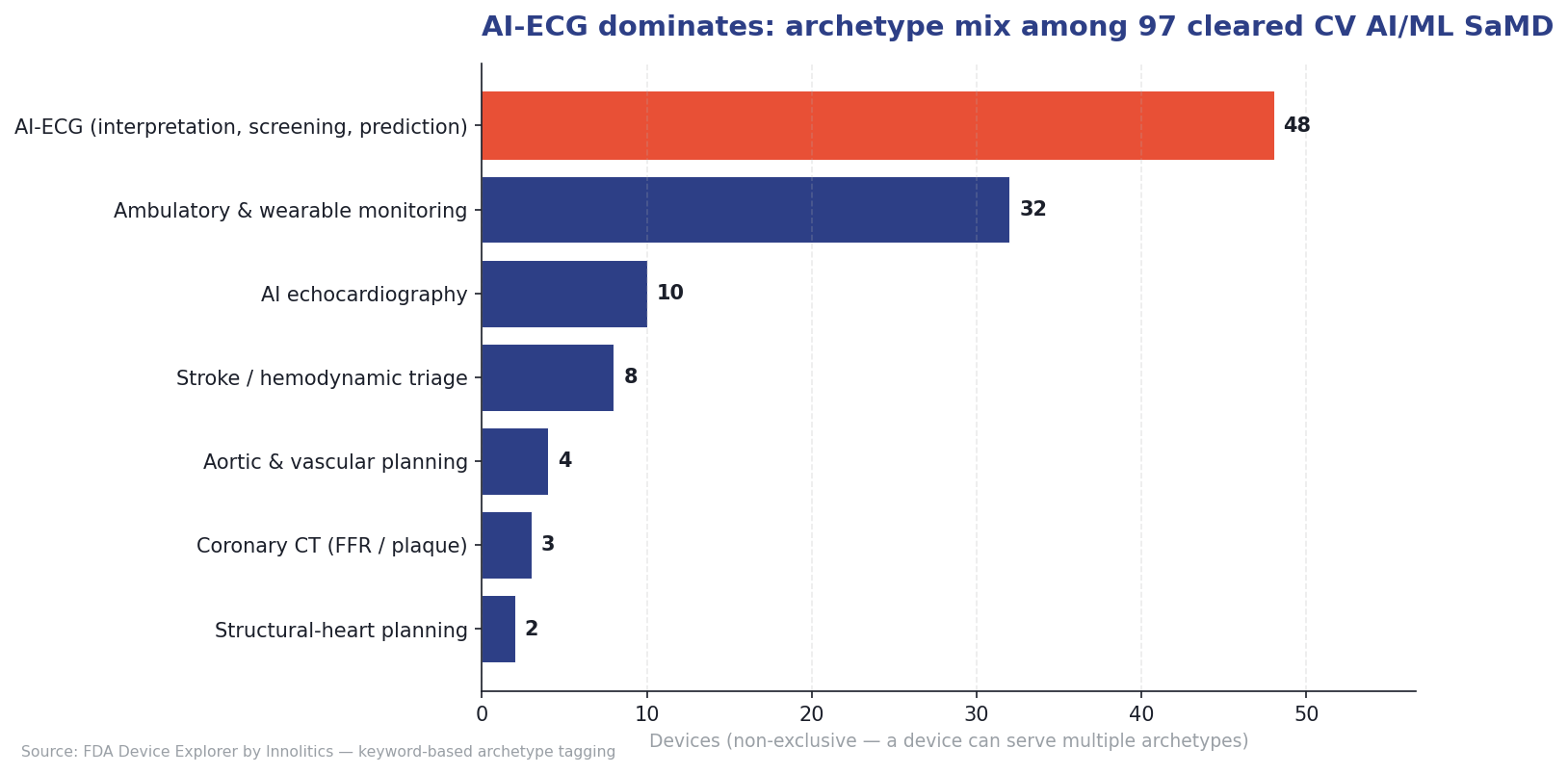
Pillar 1: AI-ECG (48 of 97 devices) 🔗
The ECG is the most common diagnostic test in medicine and the cheapest, most standardized, most easily archived signal in cardiology. AI-ECG accordingly dominates the corpus: 48 of the 97 cleared devices (50%) apply machine learning to ECG waveforms. The pillar splits into three sub-archetypes.
The first is automated interpretation, devices that replace or accelerate a human ECG over-reader. Examples include AliveCor's Corvair Monza (K252589, 2026), which produces an initial automated read of resting twelve-lead ECGs for clinician review; Cardiologs' Holter Platform (K250569, 2025); and Medicalgorithmics' ambulatory analyzers (four clearances). These typically clear under the established DPS (Holter ECG analysis software) and MHX (diagnostic 12-lead interpretation) codes. They are evolutions of decades-old software, with deep learning replacing rule-based signal processing. The novelty is the algorithm, not the indication, and most clear in under 200 days because predicates and validation methods are well understood.
The second is arrhythmia screening from reduced-lead and OTC devices, where consumer electronics has the deepest footprint. The QDA code (OTC electrocardiograph software) accounts for nine clearances, including Apple's ECG App, Withings ECG App (K240795), and Samsung's smartwatch ECG features. The QDB code (OTC PPG-based AFib history) adds five more. These devices give the patient, not the cardiologist, the first read on whether a wrist tracing looks like atrial fibrillation. They are positioned as information, not diagnosis, every OTC label states the device is not intended to provide a diagnosis, but the workflow effect has been substantial, generating patient-initiated cardiology referrals at a scale that did not exist a decade ago.
The third is the most novel: AI-ECG as a screening tool for diseases the ECG was never traditionally able to diagnose. Classical teaching holds that the ECG reveals electrical abnormalities (arrhythmias, ischemia, conduction blocks) and reveals structural disease only indirectly via coarse markers like LV hypertrophy voltage criteria. Deep-learning models have changed that. Trained on millions of paired ECG–echo and ECG–MRI records, neural networks can detect morphology that correlates with conditions invisible to the human eye in the trace.
Anumana, an Innovate-Mayo spinout, is the clearest example. It has cleared three AI-ECG screening algorithms in three years: a low-ejection-fraction (LEF) detector in 2023 (K232699), a refined LEF 12-lead algorithm in 2025 (K250652), and a pulmonary-hypertension detector in 2026 (K252360). Each is standalone software that consumes a routine 12-lead ECG and outputs a probability that the patient has structural disease warranting further imaging. The intended-use is consistently scoped ("intended to aid in earlier detection ... not intended to be a stand-alone diagnostic device"), but the implication is clear: every twelve-lead ECG in a deployed system becomes a potential opportunistic screen for currently underdiagnosed disease.
Tempus AI's ECG-AF (K233549, 2024) is a related example. It analyzes resting twelve-lead ECG recordings and outputs the probability of atrial fibrillation or flutter within the next twelve months. This is forecasting, not classification. Bunkerhill's ECG-EF (K250649) and Viz.ai's Viz HCM (DEN230003, 2023), which detects hypertrophic cardiomyopathy under its own new product code (QXO), round out the screening-from-ECG archetype.
This sub-archetype creates new clinical information from old data rather than accelerating interpretation of new data. From a payer perspective the distinction matters: a screening output from an ECG already billed does not require a new technical fee. This might mean these cardiovascular AI applications could be a stronger case for reimbursement in the future not subject to bundling.
Pillar 2: Wearable and Ambulatory PPG (32 of 97 devices) 🔗
Adjacent to AI-ECG is wearable and ambulatory monitoring, with 32 clearances. The defining feature: the device is on the patient continuously (wrist, chest patch, fingertip, hospital-room camera), and the AI runs against a continuous stream rather than a discrete tracing.
The product-code architecture is younger and more fragmented than ECG's. Relevant codes include DXH (PPG pulse-rate measurement, 4 devices), QDB (OTC PPG AFib history feature, 5 devices), QME (contactless / video vital-sign software, 4 devices), and one-off codes opened by individual De Novos including Apple's loss-of-pulse detection (SDY, K242967) and Apple's Hypertension Notification Feature (SFR, K250507).
The clinical premise is opportunistic screening: using a device the patient already wears to detect conditions before they would otherwise have presented. Apple Watch has industrialized this. Across eight cleared devices, Apple has added irregular-rhythm notification, AFib history, loss-of-pulse detection, and hypertension notification, each under a different product code, several with their own De Novo, and the most recent (HTNF, 2025) with a PCCP. Samsung holds three analogous clearances. Withings has cleared its ECG and PPG features under QDA. PreemptiveAI Clinical SDK (K250233) and Oxehealth's contactless camera-based vital-signs system (K251200) round out a small but growing set of non-wrist-worn ambulatory tools.
The key regulatory feature is the OTC carve-out. A large share of the wearable/ambulatory devices are OTC, sold and used without a prescription, which is rare in AI/ML SaMD overall. It shapes the segment's commercial logic: when the regulated party is the patient rather than a prescribing clinician, the device's job is not to diagnose but to prompt the patient to seek diagnosis. Every OTC label includes a "not intended to provide a diagnosis" statement that functions as both a regulatory boundary and a liability shield.
What the wearable pillar has not produced is also worth noting. There are no cleared wearable AI devices for continuous LV function monitoring, early heart-failure decompensation detection, or multi-modal physiological forecasting (e.g., "you are about to have an MI" notifications). Several startups are pursuing each, but none have cleared. The market still treats wearable AI primarily as arrhythmia screening and vital-signs measurement.
Pillar 3: AI Echocardiography (10 of 97 devices) 🔗
Echocardiography is cardiology's most operator-dependent imaging modality. Acquiring a diagnostic-quality parasternal long-axis (PLAX) view requires skill, and interpretation requires standardized measurements of chamber size, wall thickness, and ejection fraction that vary substantially across readers. AI is being deployed to address both; the result is a small but rapidly growing archetype of ten cleared devices.
The leader is Ultromics, with three clearances across variants of its EchoGo platform. iCardio.ai cleared CardioVision (K251293) in 2025, operating on a single PLAX view in outpatient settings and targeting primary-care POCUS. Us2.ai cleared Us2.ca (K250151) in 2025 for cardiac-amyloidosis screening on transthoracic echocardiograms in adults with increased LV wall thickness, an example of AI targeting a specific, often-missed disease rather than generic measurement. Bunkerhill's EF-from-echo and several smaller players round out the pillar.
This pillar sits at the boundary between cardiology AI and radiology AI. The product codes (QUO, SDJ, and others) are housed in the Cardiovascular review panel, but the regulatory artifacts (DICOM-based validation, ROI-driven workflows, MRMC reader studies) look like radiology's. Expect it to grow faster than AI-ECG over the next three years as ultrasound POCUS expands into primary care, emergency medicine, and anesthesiology, and as image-quality standardization lowers the operator-dependence ceiling that has historically held echocardiography back as a mass-screening tool.
Pillar 4: Coronary CT, FFR, and Plaque Analysis (3 of 97 devices) 🔗
The smallest pillar by device count but likely the highest-revenue per device, CCTA is anchored by HeartFlow and Cleerly. HeartFlow Analysis (K213857) provides AI-based quantitative analysis of coronary CT data, including FFR-CT, a non-invasive surrogate for invasive fractional flow reserve. Cleerly ISCHEMIA (K231335) is a parallel diagnostic aid built on Cleerly's plaque-quantification software. There are only three cleared CCTA devices because the archetype is held by a few deeply integrated platforms.
This pillar is a counterpoint to AI-ECG and wearable: not all cardiology AI lives in cheap, high-volume ambulatory data. There is also a niche for AI on expensive, advanced imaging, dominated by a few companies that have built the workflow integration, clinical evidence, and reimbursement story to make CCTA-based AI commercially viable. It most resembles the radiology cohort, and that is also where category-defining CPT codes (e.g., HeartFlow's FFR-CT codes) have driven adoption.
Adjacents: Stroke and Hemodynamic Triage, Aortic and Vascular Planning, Structural-Heart Planning 🔗
Three smaller archetypes are worth flagging. Stroke and hemodynamic triage covers eight devices, including Edwards Lifesciences' Acumen HPI, Acumen Assisted Fluid Management, and Acumen Hypertension Prediction Index, plus the eCART deterioration-prediction suite and the CLEWICU System. These live primarily in the OR, ICU, and ward and forecast acute decompensation minutes to hours ahead. They are the highest-stakes devices in the cohort and tend to draw the longest review times.
Aortic and vascular planning covers four devices focused on pre-procedural sizing and planning. Structural-heart planning is the smallest with two devices but is positioned to grow as transcatheter procedures (TAVR, TEER, LAA closure) expand and AI becomes the standard tool for valve and annulus sizing.
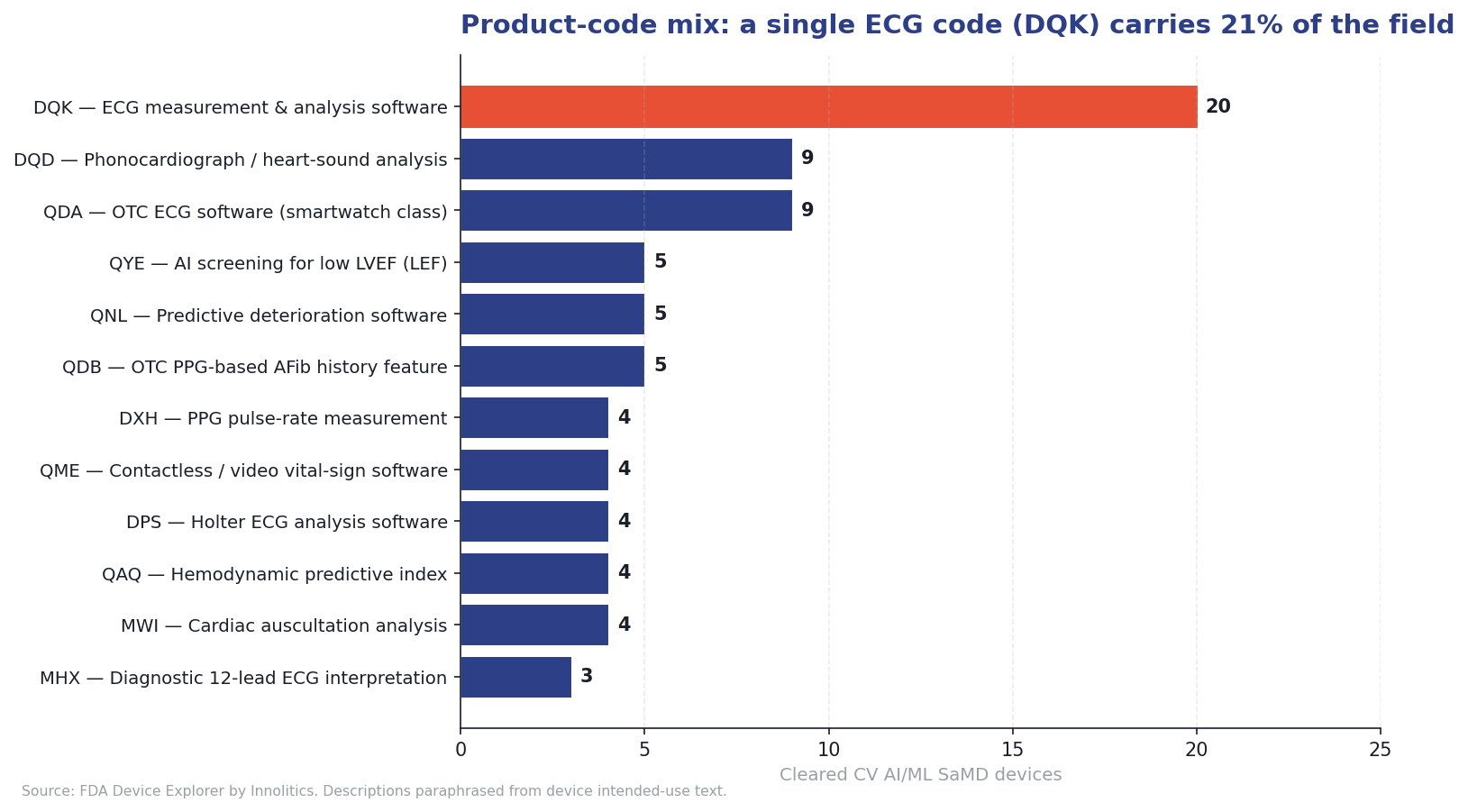
Product-code distribution reflects the archetype mix. A single ECG code, DQK (electrocardiograph measurement and analysis software), accounts for 20 of the 97 devices (21%). That is high concentration: in radiology AI, no single code carries more than about 12%. The implication is practical, if you can find a credible DQK predicate, your clearance path is much shorter than a De Novo into a new code.
The Regulatory Reality: Why Cardiology AI Is Slower Than Radiology AI 🔗
The cohort tells a consistent story about how FDA reviews cardiology AI: more slowly, and with more interest in change control, than any other specialty.
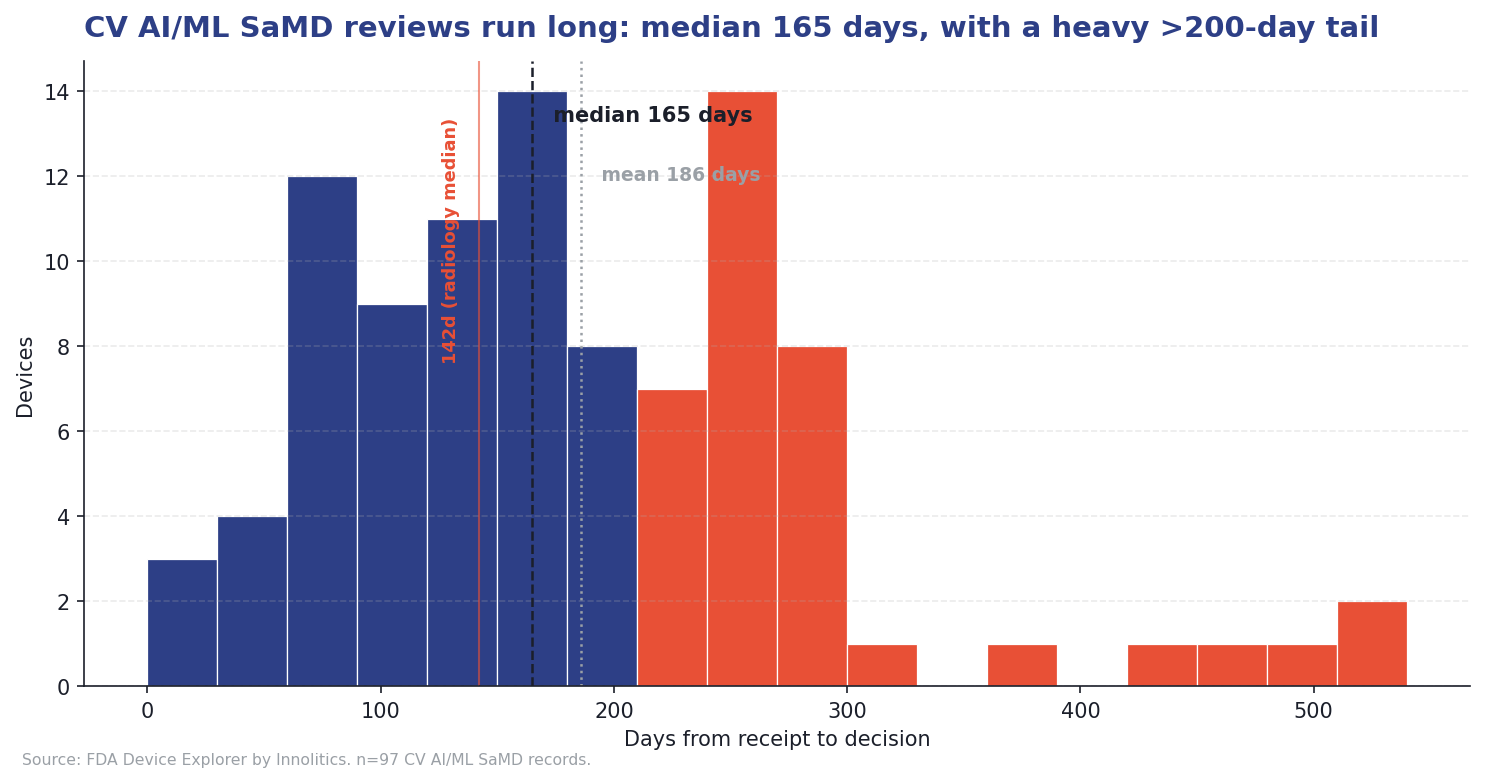
Median review time across the 97 devices is 165 days; the mean is 186 days. The fastest was 23 days; the slowest 536. Forty of the 97 (41%) took more than 200 days. The 2025 year-in-review reported a radiology AI median of about 142 days, so cardiology AI takes roughly 16% longer on average.
This reflects the structural newness of the devices, not FDA inefficiency. Two factors drive the longer reviews.
First, the cohort relies heavily on De Novo classification: eight De Novos appear (one per twelve clearances), including Apple's ECG App and Irregular Rhythm Notification, Edwards' HPI, and Viz.ai's HCM detector. Each required FDA to articulate a new classification, define special controls, and negotiate validation expectations from scratch. Subsequent 510(k)s into the resulting codes (QDA, QDB, QAQ, QXO) are faster, but the De Novo itself is long and expensive.
Second, more devices are predictive rather than purely diagnostic. Edwards' HPI, Tempus' ECG-AF, the eCART deterioration suite, and Anumana's screening algorithms make claims about future events, and validating future-event claims requires substantially more clinical evidence than present-state classification. FDA reviews these submissions more cautiously, and the long tail reflects that.
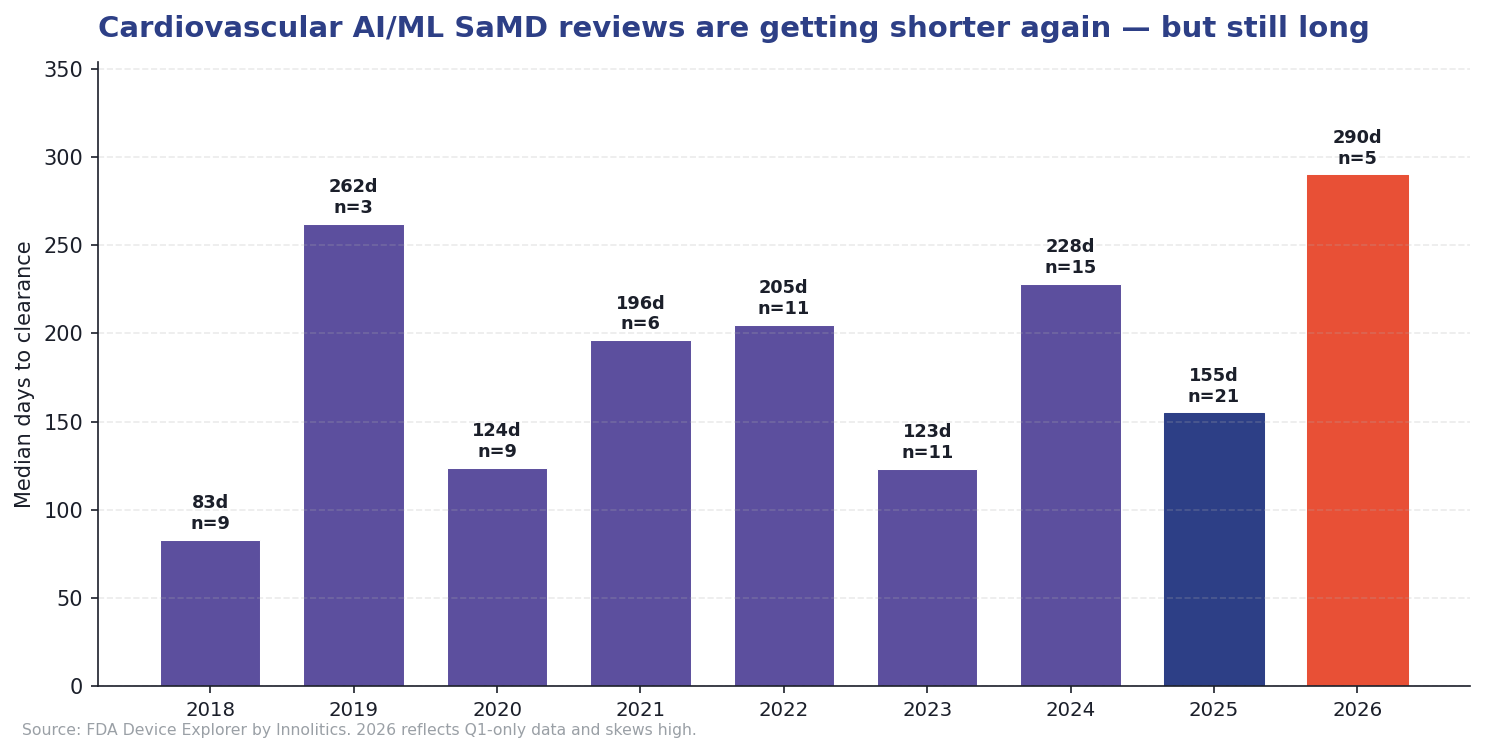
The trend is improving. After peaking at a median of 228 days in 2024, review time dropped to 155 days in 2025, close to radiology AI. The early-2026 spike to 290 days is a small-sample artifact (n=5) weighted by complex submissions clearing after long reviews and should not be read as a regression. Volume-weighted, the longer-run trajectory is downward. As predicates for the new codes accumulate, cardiology review times should continue to converge toward the radiology median.
The PCCP Adoption Curve 🔗
An interesting regulatory development in cardiology AI is the moderate adoption of the Predetermined Change Control Plan (PCCP). A PCCP is a section of a 510(k) or De Novo submission in which the manufacturer pre-specifies the post-market algorithm changes allowed without a new submission.
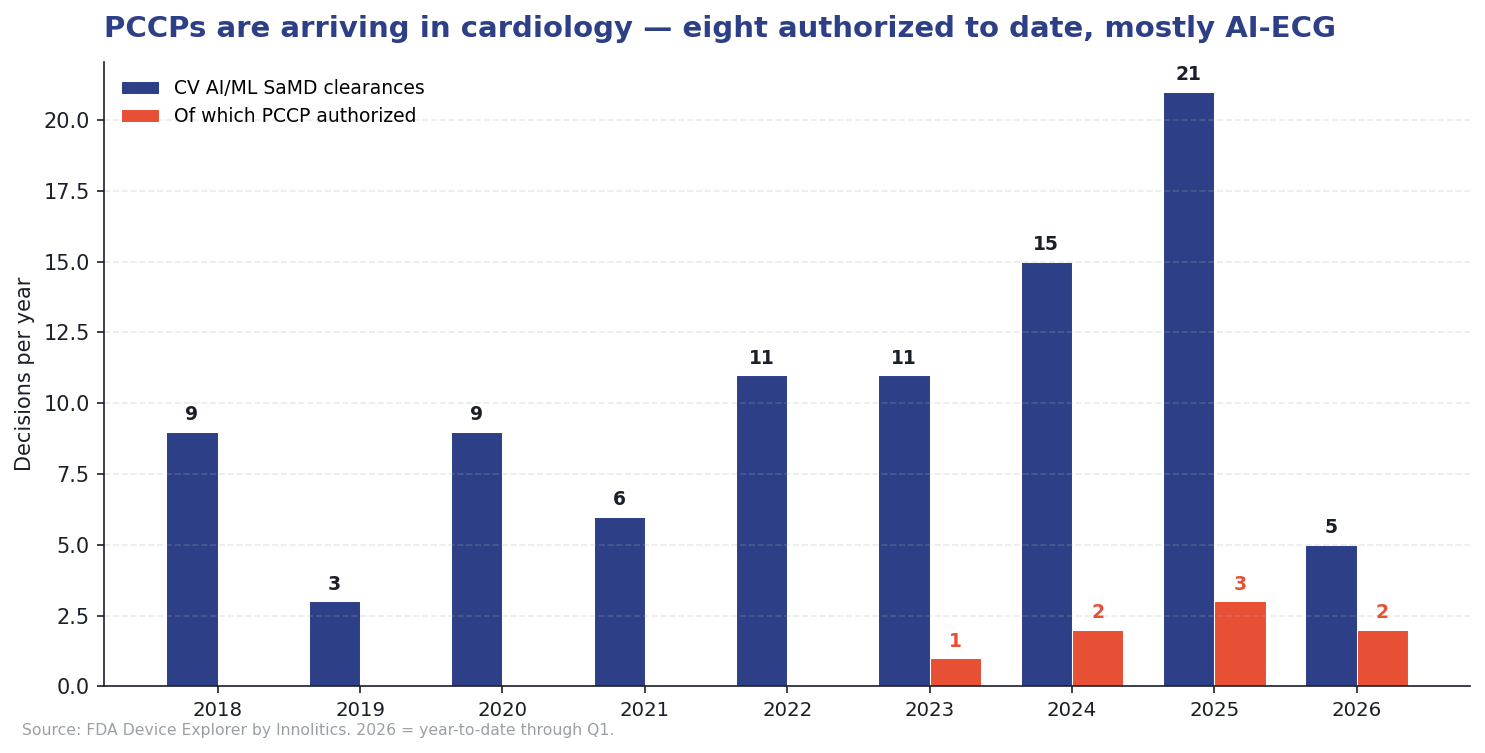
Eight of the 97 cleared devices include an authorized PCCP, 8.2% of the cohort, but all eight have been authorized in the last 30 months. The first appeared in September 2023 (Anumana's Low Ejection Fraction AI-ECG Algorithm, K232699). By Q1 2026, two of the most recent five clearances include a PCCP, and the trailing-twelve-month rate is roughly one in five.

Of the eight cardiology PCCPs, six are for AI-ECG or AI-PPG algorithms:
| # | Year | Manufacturer | Device | Archetype |
|---|---|---|---|---|
| 1 | 2023 | Anumana | Low Ejection Fraction AI-ECG Algorithm (K232699) | AI-ECG screening |
| 2 | 2024 | Clew Medical | CLEWICU System (K233216) | ICU triage |
| 3 | 2024 | AliveCor | Corvair (K231010) | AI-ECG interpretation |
| 4 | 2025 | Anumana | ECG-AI LEF 12-lead (K250652) | AI-ECG screening |
| 5 | 2025 | Apple | Hypertension Notification Feature (K250507) | Wearable PPG |
| 6 | 2025 | Csd Labs | eMurmur Heart AI (K252284) | Phonocardiogram |
| 7 | 2026 | AliveCor | Corvair Monza (K252589) | AI-ECG interpretation |
| 8 | 2026 | Anumana | ECG-AI Pulmonary Hypertension 12-lead (K252360) | AI-ECG screening |
This concentration is not coincidental. Waveform-based AI fits PCCP well: training data is plentiful and curatable, architectures are stable, and the changes manufacturers want to make post-market (retraining on new demographics, refining thresholds, expanding lead configurations) are exactly what PCCP is designed to handle. The relative scarcity of PCCPs in radiology AI suggests imaging is still working out its post-market change processes; cardiology has effectively standardized on PCCP for waveform AI.
Anumana's three PCCPs are among the largest PCCP portfolios in any specialty in the FDA database. AliveCor's two are second. Apple's PCCP for the Hypertension Notification Feature is an important precedent: it shows PCCPs work for OTC consumer-facing screening features, not just prescription algorithms, a more difficult case because the "user" is the patient and change control runs at consumer scale.
What This Means for Builders of Cardiology AI 🔗
The data supports a small set of strategic conclusions for anyone building, investing in, or buying cardiology AI/ML SaMD.
- Cardiology AI is mostly a signals market, not an imaging-heavy one. AI-ECG and wearable PPG account for 80 of the 97 cleared devices, and most near-term growth sits in those pillars. Build the data pipelines, evidence, and post-market workflow around continuous waveforms first; treat imaging as complementary.
- The OTC consumer-electronics tier is real and durable, and it is mostly Apple, Samsung, and Withings under the QDA/QDB codes they opened. Retail distribution, "information, not diagnosis" labeling, and prospective validation make it a hard tier to enter directly. Most startups are better off building prescription software that consumes OTC outputs.
- The third generation of AI-ECG is already here, and the runway is long. Gen 1 was automated interpretation, Gen 2 was reduced-lead arrhythmia detection, Gen 3 is screening for diseases the ECG was never traditionally able to detect (Anumana, Tempus, Bunkerhill, Viz.ai). Expect more competitors as health systems treat their ECG archives as a screening asset rather than a recordkeeping one.
- AI echo is the next archetype to scale. Primary-care POCUS dynamics that lifted AI-ECG screening are now lifting AI echo; expect a sharp rise in QUO and SDJ clearances for amyloidosis, HFpEF, and structural-heart planning. Winners will standardize acquisition as well as interpretation, because the bottleneck in primary-care echo is the scan, not the read.
Conclusion 🔗
For most of the last decade, cardiovascular AI has trailed radiology in attention. The FDA's data shows that has changed. The cardiology cohort is growing faster on a percentage basis than radiology; it is the dominant home of PCCP; it is the only specialty where consumer-electronics companies have built top-tier AI/ML SaMD portfolios; and it is the clearest example of AI making an existing test detect diseases it was never designed to find. It is also where the pivot from "AI helps the specialist read faster" to "AI creates a new screening tier in front of the specialist" has gone furthest.
Don't skip the cardiology section in the next year-in-review. The most important architectural shifts in clinical AI are happening there first, on signals rather than pixels. The Apple Watch on a patient's wrist, the twelve-lead ECG in a primary-care clinic, and the arterial line in the OR are each gaining AI augmentation faster than any other modality in cardiology.
References 🔗
[1] FDA Device Explorer by Innolitics. Cardiovascular AI/ML SaMD Clearances, 2006–2026 YTD. Data accessed April 2026. https://fda.innolitics.com/
[2] Innolitics. Year in Review: AI/ML Medical Device 510(k) Clearances (2025). https://innolitics.com/articles/year-in-review-ai-ml-medical-device-k-clearances/
[3] U.S. Food and Drug Administration. Marketing Submission Recommendations for a Predetermined Change Control Plan for Artificial Intelligence-Enabled Device Software Functions. Final Guidance, December 2024. https://www.fda.gov/regulatory-information/search-fda-guidance-documents/marketing-submission-recommendations-predetermined-change-control-plan-artificial-intelligence
###
The takeaway for builders: cardiology AI has the longest regulatory path, the newest pathways, and the heaviest post-market expectations (PCCP, real-world performance monitoring, change control) of any AI/ML SaMD specialty. A 16% longer median review time, heavy De Novo reliance, and near-mandatory PCCP for waveform AI cannot be engineered around late. They need to be in the regulatory strategy from day one.
Innolitics has been doing this work since 2012. We are a team of physicians, software engineers, and regulatory experts focused on AI/ML SaMD, with 50+ cleared FDA submissions including AI-ECG screening algorithms, AI echocardiography tools, plaque-analysis CCTA platforms, and consumer-grade arrhythmia features. We are also the only firm that puts a speed and certainty guarantee on its core offerings:
- Guaranteed AI/ML SaMD 510(k), our flagship Fast 510(k) service: submission in 3 months and FDA clearance in 9 months total, or a partial refund.
- Guaranteed Regulatory Strategy, Q-Sub, and BDD in 2 weeks, to lock in the path of least resistance and unlock expedited review where the device qualifies.
- Guaranteed Breakthrough Device Designation, for cardiology devices targeting life-threatening or irreversibly debilitating conditions.
- Guaranteed QMS in 2 months, so your post-market PCCP commitments are backed by an actual quality system.
- Guided 510(k), for teams that want to own the documentation but need expert review at every sprint.
If you are building in any of the four pillars, AI-ECG, wearable PPG, AI echo, or CCTA, and want a partner who has already mapped the regulatory terrain, let's meet. The 98th device in this dataset could be yours.